UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
SCHEDULE 14D-9
(Rule 14d-101)
SOLICITATION/RECOMMENDATION STATEMENT
UNDER SECTION 14(d)(4) OF THE SECURITIES EXCHANGE ACT OF 1934
Blueprint Medicines Corporation
(Name of Subject Company)
Blueprint Medicines Corporation
(Name of Person(s) Filing Statement)
COMMON STOCK, PAR VALUE $0.001 PER SHARE
(Title of Class of Securities)
09627Y109
(CUSIP Number of Class of Securities)
Kathryn Haviland
President and Chief Executive Officer
Blueprint Medicines Corporation
45 Sidney Street
Cambridge, Massachusetts 02139
(617) 374-7580
With copies to:
Stuart M. Cable, Esq.
Lisa R. Haddad, Esq.
James Ding, Esq.
Goodwin Procter LLP
100 Northern Avenue
Boston, Massachusetts 02210
(617) 570-1000
(Name, address, and telephone numbers of person authorized to receive notices and communications
on behalf of the persons filing statement)
| x | Check the box if the filing relates solely to preliminary communications made before the commencement of a tender offer. |
This Schedule 14D-9 filing relates solely to preliminary communications made before the commencement of a planned tender offer (the “Offer”) by Rothko Merger Sub, Inc., a Delaware corporation (“Purchaser”) and a wholly owned subsidiary of Aventis Inc., a Pennsylvania corporation (“Aventis”) and wholly owned subsidiary of Sanofi, a French société anonyme (“Parent”), to acquire all of the issued and outstanding shares of common stock, par value $0.001 per share, of Blueprint Medicines Corporation, a Delaware corporation (the “Company” or “Blueprint”), to be commenced pursuant to the Agreement and Plan of Merger, dated as of June 2, 2025, by and among Parent, Aventis, Purchaser and the Company (the “Merger Agreement”). Upon the consummation of the Offer, Purchaser will merge with and into the Company (the “Merger”) pursuant to Section 251(h) of the Delaware General Corporation Law, with the Company as the surviving corporation. This Schedule 14D-9 filing consists of the following documents relating to the proposed Offer and Merger:
| · | Exhibit 99.1: Email from Kathryn Haviland, President and Chief Executive Officer of the Company, to employees on June 2, 2025. |
| · | Exhibit 99.2: Blueprint Employee FAQ, first used on June 2, 2025. |
| · | Exhibit 99.3: Posting by the Company via LinkedIn and X on June 2, 2025. |
| · | Exhibit 99.4: Investor Relations Message, dated June 2, 2025. |
| · | Exhibit 99.5: Blueprint Medicines + Sanofi Overview for the Blue Crew dated June 2, 2025. |
Cautionary Note Regarding Forward-Looking Statements
This document contains forward-looking statements regarding, among other things, the proposed acquisition of the Company by Parent, the expected timetable for completing the transaction, and the Company’s future financial or operating performance. The Company generally identifies forward-looking statements by terminology such as “aim,” “may,” “will,” “could,” “would,” “should,” “expect,” “plan,” “anticipate,” “intend,” “believe,” “estimate,” “predict,” “project,” “opportunity,” “contemplate,” “potential,” “continue,” “target” or the negative of these terms or other similar words, although not all forward-looking statements contain such terms. All statements that are not statements of historical facts are, or may be deemed to be, forward-looking statements. These forward-looking statements are only predictions, and such statements are based on current expectations and projections about future events and trends as well as the beliefs and assumptions of management. Forward-looking statements are subject to a number of risks and uncertainties, many of which involve factors or circumstances that are beyond the Company’s control. Actual results could differ materially from those stated or implied in forward-looking statements due to a number of factors, including but not limited to: (i) risks associated with the timing of the closing of the proposed transaction, including the risks that a condition to closing would not be satisfied within the expected timeframe or at all or that the closing of the proposed transaction will not occur; (ii) uncertainties as to how many of the Company’s stockholders will tender their shares in the offer; (iii) the possibility that a governmental entity may prohibit, delay or refuse to grant approval for the consummation of the transaction; (iv) the possibility that competing offers will be made; (v) the occurrence of any event, change or other circumstance that could give rise to the termination of the transaction; (vi) the outcome of any legal proceedings that may be instituted against the parties and others related to the merger agreement; (vii) unanticipated difficulties or expenditures relating to the proposed transaction, the response of business partners and competitors to the announcement of the proposed transaction, and/or potential difficulties in employee retention as a result of the announcement and pendency of the proposed transaction; (viii) risks related to non-achievement of the CVR milestones and that holders of the CVRs will not receive payments in respect of the CVRs; (ix) the risk that the marketing and sale of AYVAKIT/ AYVAKYT or any future approved drugs may be unsuccessful or less successful than anticipated, or that AYVAKIT/ AYVAKYT may not gain market acceptance by physicians, patients, third-party payors and others in the medical community; (x) the risk that the market opportunities for AYVAKIT/ AYVAKYT or the Company’s drug candidates are smaller than the Company estimates or that any approval that the Company obtains may be based on a narrower definition of the patient population that the Company anticipates; (xi) the risk of delay of any current or planned clinical trials or the development of the Company’s current or future drug candidates, including but not limited to BLU-808 and elenestinib; (xii) risks related to the Company’s ability to successfully demonstrate the safety and efficacy of its drug candidates and gain approval of its drug candidates on a timely basis, if at all; (xiii) preclinical and clinical results for the Company’s drug candidates may not support further development of such drug candidates either as monotherapies or in combination with other agents or may impact the anticipated timing of data or regulatory submissions; (xiv) the timing of the initiation of clinical trials and trial cohorts at clinical trial sites and patient enrollment rates may be delayed or slower than anticipated; (xv) actions of regulatory agencies may affect the Company’s approved drugs or its current or future drug candidates, including affecting the initiation, timing and progress of clinical trials, as well as the pricing of its drug candidates; (xvi) risks related to the Company’s ability to obtain, maintain and enforce patent and other intellectual property protection for its products and current or future drug candidates it is developing; (xvii) the success of the Company’s current and future collaborations, financing arrangements, partnerships or licensing and other arrangements; (xviii) risks related to the Company’s liquidity and financial position and the accuracy of its estimates of revenues, expenses, cash burn, and capital requirements; and (xix) those risks detailed in the Company’s most recent Annual Report on Form 10-K and subsequent reports filed with the SEC, as well as other documents that may be filed by the Company from time to time with the SEC. The Company cannot assure you that the events and circumstances reflected in the forward-looking statements will be achieved or occur, and actual results could differ materially from those projected in the forward-looking statements. The forward-looking statements made in this document relate only to events as of the date on which the statements are made. The Company undertakes no obligation to update any forward-looking statement to reflect events or circumstances after the date on which the statement is made or to reflect the occurrence of unanticipated events.
Additional Information and Where to Find It
The tender offer referred to in this document has not yet commenced. This document is for informational purposes only and is neither an offer to purchase nor a solicitation of an offer to sell shares, nor is it a substitute for the tender offer materials that Parent and its acquisition subsidiary will file with the SEC upon commencement of the tender offer. At the time the tender offer is commenced, Parent and its acquisition subsidiary will cause to be filed a tender offer statement on Schedule TO with the SEC, and the Company will file a solicitation/recommendation statement on Schedule 14D-9 with respect to the tender offer. THE TENDER OFFER STATEMENT (INCLUDING AN OFFER TO PURCHASE, A RELATED LETTER OF TRANSMITTAL AND OTHER OFFER DOCUMENTS) AND THE SOLICITATION/RECOMMENDATION STATEMENT WILL CONTAIN IMPORTANT INFORMATION THAT SHOULD BE READ CAREFULLY AND CONSIDERED BY THE COMPANY’S STOCKHOLDERS BEFORE ANY DECISION IS MADE WITH RESPECT TO THE TENDER OFFER. Both the tender offer statement and the solicitation/recommendation statement will be mailed to the Company’s stockholders free of charge. A free copy of the tender offer statement and the solicitation/recommendation statement will also be made available to all stockholders of the Company by accessing www.blueprintmedicines.com or by contacting the Company’s Investor Relations contact at ir@blueprintmedicines.com. In addition, the tender offer statement and the solicitation/recommendation statement (and all other documents filed with the SEC) will be available at no charge on the SEC’s website: www.sec.gov, upon filing with the SEC.
THE COMPANY’S STOCKHOLDERS ARE ADVISED TO READ THE SCHEDULE TO AND THE SCHEDULE 14D-9, AS EACH MAY BE AMENDED OR SUPPLEMENTED FROM TIME TO TIME, AND ANY OTHER RELEVANT DOCUMENTS FILED WITH THE SEC WHEN THEY BECOME AVAILABLE BEFORE THEY MAKE ANY DECISION WITH RESPECT TO THE TENDER OFFER, BECAUSE THEY WILL CONTAIN IMPORTANT INFORMATION ABOUT THE PROPOSED TRANSACTION AND THE PARTIES THERETO.
Exhibit 99.1
CEO email message to Blueprint employees
E-mail Subject: Blueprint and Sanofi announce combination to accelerate leadership in immunology
From: Kate Haviland
To: All Blueprint employees and contractors
Confidential – Internal Use Only
Blue Crew,
Since our inception, Blueprint has worked towards our singular mission to alleviate human suffering through innovative and life-changing medicines. We have always measured our success in the number of people around the world whose lives are better, healthier and easier because of the work we do.
And we are delivering on our mission with AYVAKIT®/AYVAKYT® (avapritinib), which is the culmination of more than a decade of pioneering research in systemic mastocytosis (SM). This medicine – that we invented – is now having a profound and meaningful impact on the lives of thousands of patients with SM. Across our industry, this achievement is rare, and I’m incredibly proud of our work together to build a special company that is positively impacting the world.
Today, we announced that we have reached an agreement to join Sanofi (see attached press release), as we work together to bring the innovation we are driving across our business to more patients around the world. Sanofi has the resources and reach to make our vision a reality faster and at a larger scale. As many of you know, Sanofi is a global leader in rare diseases and immunology, and has a long history of serving patients around the world and solving important medical needs at scale.
This transaction is an exceptional recognition of the value we collectively have created for patients with the research, development and global commercialization of AYVAKIT, as well as our R&D pipeline, including elenestinib and BLU-808. This next chapter holds the potential to amplify our mission, not change it – and what I ask today is that we all remain grounded in the purpose that brought us here.
As we all process this news, I know that the change it represents will bring forward a range of feelings and questions. That is okay and we will meet this moment together, with honesty, optimism, and empathy, as we always have. I believe this change creates space for new possibilities and a chance to unlock untapped potential.
At Blueprint, it's in our DNA to pioneer new ground. Time and again, as a team we have turned uncharted territory into deep expertise. We have managed significant ambiguity and relentless change to build a truly distinctive company that has and continues to deliver meaningful value. All our effort has led to this very moment of recognition and opportunity.
So what happens now?
What happens next is a continued commitment to transparency, collaboration, and shared understanding – and your voice is an essential part of that. I invite you to join a company meeting later today at 10:00 am ET / 4:00 pm CET where we’ll share more, answer the questions we can, and navigate this next step together. Please keep an eye out for the calendar invitation this morning.
In addition, later today we will post a dedicated page on Blink with a repository of information, including FAQs for employees as well as approved messages to help you respond to any inquiries from your external stakeholders including customers, business partners and vendors, if needed. Over time, we will update this content when needed, with a commitment to communicate transparently.
Until the transaction officially closes – anticipated in the third quarter of 2025 – both companies will continue to operate independently, guided by our existing leadership and structure. During this time, we must continue to focus on our current priorities and day-to-day responsibilities to meet our commitments to the patients and healthcare providers who rely on us, and to set the foundation for the work we will do together with Sanofi in the future. Our values are deeply rooted and will continue to ground us as we focus on what matters most: delivering on our commitments, making a meaningful difference in the lives of patients, and supporting each other’s wellbeing.
If in the coming weeks you receive any inquiries from investors or media, please direct them to Jim Baker, SVP Corporate Affairs. In addition, we ask that all Blueprint employees refrain from any social media postings about the transaction until closing.
I am deeply grateful to have the opportunity to work with such an incredible group of colleagues, who challenge me, who I learn something new from every day, who help me think differently, who make me laugh, and who have shown up day after day to do the hard work. The work you do matters and most importantly, it matters to our patients and their families which is simply captured in the opening from a letter we received from a patient which said, “To my Heros at Blueprint Medicines.”
As we step into this next chapter, let’s stay grounded in who we are, open to what’s possible, and committed to moving forward with our patient-focused purpose. See you later today.
All the best,
Kate
Kate Haviland
President and Chief Executive Officer
Blueprint Medicines
Additional Information about the Proposed Transaction and Where to Find It
The tender offer referred to in this communication has not yet commenced. This communication is for informational purposes only and is neither an offer to purchase nor a solicitation of an offer to sell shares, nor is it a substitute for the tender offer materials that Sanofi and its acquisition subsidiary will file with the U.S. Securities and Exchange Commission (the “SEC”) upon commencement of the tender offer. At the time the tender offer is commenced, Sanofi and its acquisition subsidiary will cause to be filed a tender offer statement on Schedule TO with the SEC, and Blueprint Medicines Corporation (the “Company”) will file a solicitation/recommendation statement on Schedule 14D-9 with respect to the tender offer. THE TENDER OFFER STATEMENT (INCLUDING AN OFFER TO PURCHASE, A RELATED LETTER OF TRANSMITTAL AND OTHER OFFER DOCUMENTS) AND THE SOLICITATION/RECOMMENDATION STATEMENT WILL CONTAIN IMPORTANT INFORMATION THAT SHOULD BE READ CAREFULLY AND CONSIDERED BY THE COMPANY’S STOCKHOLDERS BEFORE ANY DECISION IS MADE WITH RESPECT TO THE TENDER OFFER. Both the tender offer statement and the solicitation/recommendation statement will be mailed to the Company’s stockholders free of charge. A free copy of the tender offer statement and the solicitation/recommendation statement will also be made available to all stockholders of the Company by accessing www.blueprintmedicines.com or by contacting the Company’s Investor Relations contact at ir@blueprintmedicines.com. In addition, the tender offer statement and the solicitation/recommendation statement (and all other documents filed with the SEC) will be available at no charge on the SEC’s website: www.sec.gov, upon filing with the SEC.
THE COMPANY’S STOCKHOLDERS ARE ADVISED TO READ THE SCHEDULE TO AND THE SCHEDULE 14D-9, AS EACH MAY BE AMENDED OR SUPPLEMENTED FROM TIME TO TIME, AND ANY OTHER RELEVANT DOCUMENTS FILED WITH THE SEC WHEN THEY BECOME AVAILABLE BEFORE THEY MAKE ANY DECISION WITH RESPECT TO THE TENDER OFFER, BECAUSE THEY WILL CONTAIN IMPORTANT INFORMATION ABOUT THE PROPOSED TRANSACTION AND THE PARTIES THERETO.
Cautionary Statement Regarding Forward-Looking Statements
This communication contains forward-looking statements regarding, among other things, the proposed acquisition of the Company by Sanofi, the expected timetable for completing the transaction, and the Company’s future financial or operating performance. The Company generally identifies forward-looking statements by terminology such as “aim,” “may,” “will,” “could,” “would,” “should,” “expect,” “plan,” “anticipate,” “intend,” “believe,” “estimate,” “predict,” “project,” “opportunity,” “contemplate,” “potential,” “continue,” “target” or the negative of these terms or other similar words, although not all forward-looking statements contain such terms. All statements that are not statements of historical facts are, or may be deemed to be, forward-looking statements. These forward-looking statements are only predictions, and such statements are based on current expectations and projections about future events and trends as well as the beliefs and assumptions of management. Forward-looking statements are subject to a number of risks and uncertainties, many of which involve factors or circumstances that are beyond the Company’s control. Actual results could differ materially from those stated or implied in forward-looking statements due to a number of factors, including but not limited to: (i) risks associated with the timing of the closing of the proposed transaction, including the risks that a condition to closing would not be satisfied within the expected timeframe or at all or that the closing of the proposed transaction will not occur; (ii) uncertainties as to how many of the Company’s stockholders will tender their shares in the offer; (iii) the possibility that a governmental entity may prohibit, delay or refuse to grant approval for the consummation of the transaction; (iv) the possibility that competing offers will be made; (v) the occurrence of any event, change or other circumstance that could give rise to the termination of the transaction; (vi) the outcome of any legal proceedings that may be instituted against the parties and others related to the merger agreement; (vii) unanticipated difficulties or expenditures relating to the proposed transaction, the response of business partners and competitors to the announcement of the proposed transaction, and/or potential difficulties in employee retention as a result of the announcement and pendency of the proposed transaction; (viii) risks related to non-achievement of the contingent value right (“CVR”) milestones and that holders of the CVRs will not receive payments in respect of the CVRs; (ix) the risk that the marketing and sale of AYVAKIT/ AYVAKYT or any future approved drugs may be unsuccessful or less successful than anticipated, or that AYVAKIT/ AYVAKYT may not gain market acceptance by physicians, patients, third-party payors and others in the medical community; (x) the risk that the market opportunities for AYVAKIT/ AYVAKYT or the Company’s drug candidates are smaller than the Company estimates or that any approval that the Company obtains may be based on a narrower definition of the patient population that the Company anticipates; (xi) the risk of delay of any current or planned clinical trials or the development of the Company’s current or future drug candidates, including but not limited to BLU-808 and elenestinib; (xii) risks related to the Company’s ability to successfully demonstrate the safety and efficacy of its drug candidates and gain approval of its drug candidates on a timely basis, if at all; (xiii) preclinical and clinical results for the Company’s drug candidates may not support further development of such drug candidates either as monotherapies or in combination with other agents or may impact the anticipated timing of data or regulatory submissions; (xiv) the timing of the initiation of clinical trials and trial cohorts at clinical trial sites and patient enrollment rates may be delayed or slower than anticipated; (xv) actions of regulatory agencies may affect the Company’s approved drugs or its current or future drug candidates, including affecting the initiation, timing and progress of clinical trials, as well as the pricing of its drug candidates; (xvi) risks related to the Company’s ability to obtain, maintain and enforce patent and other intellectual property protection for its products and current or future drug candidates it is developing; (xvii) the success of the Company’s current and future collaborations, financing arrangements, partnerships or licensing and other arrangements; (xviii) risks related to the Company’s liquidity and financial position and the accuracy of its estimates of revenues, expenses, cash burn, and capital requirements; and (xix) those risks detailed in the Company’s most recent Annual Report on Form 10-K and subsequent reports filed with the SEC, as well as other documents that may be filed by the Company from time to time with the SEC. The Company cannot assure you that the events and circumstances reflected in the forward-looking statements will be achieved or occur, and actual results could differ materially from those projected in the forward-looking statements. The forward-looking statements made in this communication relate only to events as of the date on which the statements are made. The Company undertakes no obligation to update any forward-looking statement to reflect events or circumstances after the date on which the statement is made or to reflect the occurrence of unanticipated events.
Exhibit 99.2
FAQ for blueprint employees
About the transaction
| 1. | What is the rationale for the transaction? |
Blueprint and Sanofi are a strong strategic fit, and we believe this complementary combination will accelerate our mission to deliver life-changing medicines to patients. Blueprint’s portfolio of mast cell targeted therapies fits seamlessly with Sanofi’s global leadership in rare diseases and immunology and inflammation. With exceptional strength in these areas spanning discovery to commercialization, Sanofi will enable us to accelerate our mission and help many more patients around the world.
| 2. | What are the transaction terms? What is a Contingent Value Right (CVR) and how does it work? |
Under the terms of the agreement, Sanofi will acquire Blueprint for approximately $9.5 billion, or up to $135 per share in total consideration. At close, each shareholder will receive $129 per share in cash, as well as one contingent value right (CVR) per share, each representing the right to receive two potential milestone payments of $2 and $4, respectively.
Contingent value rights are rights granted by an acquirer to a company’s shareholders to provide them additional value if future milestones are achieved. CVRs are often used in transactions to recognize the potential value of a development-stage asset where there is some uncertainty of clinical or regulatory success. In this case, the CVRs represent the right to receive two contingent payments if certain development and regulatory milestones for BLU-808 are achieved.
The first clinical development milestone payment is $2 per CVR, payable upon the dosing of the fifth patient in the first Phase 2b Study or the first Phase 3 Study (whichever occurs first) for BLU-808 in any indication. This milestone will need to be achieved by December 31, 2028 in order for the CVR payment to be made.
The second regulatory milestone payment is $4 per CVR, payable upon receipt from the U.S. Food and Drug Administration (FDA) of approval of a new drug application for BLU-808, that is necessary for the commercial marketing and sale of BLU-808 in the United States for any indication. This milestone will need to be achieved by June 30, 2032 in order for the CVR payment to be made.
In the event that either or both milestones are achieved, the CVR will entitle each employee, who holds a CVR on the date of close, to a cash payout of the milestone payment. It's important to note that the CVR is non-tradable, meaning it cannot be bought or sold on the market.
| 3. | Has Sanofi shared their plans for our oncology portfolio? |
The primary area of focus of our discussions with Sanofi leading to this transaction was our mast cell portfolio, including AYVAKIT, elenestinib and BLU-808, which are all highly aligned with Sanofi’s portfolio and vision for leadership in immunology and inflammation. Our oncology programs are also promising, though much earlier in development and in the preclinical setting. We expect Sanofi to assess their strategic fit and opportunities for advancement after the transaction closes.
| 4. | When will the transaction be completed? |
We expect to complete, or close, the transaction in the third quarter of 2025.
| 5. | What needs to happen before the transaction can be completed? |
The transaction is subject to customary closing conditions, including the valid tender of a majority of outstanding Blueprint shares in the related tender offer and receipt of certain regulatory approvals.
| 6. | What happens next? |
Blueprint and Sanofi will continue to operate as independent companies until the transaction closes. To fulfill our commitments to the patients, providers and partners who rely on us and to further strengthen the foundation for the work we will do with Sanofi in the future, all of us at Blueprint must continue to focus on our current priorities and day-to-day responsibilities. Until the transaction closes, we will operate our business as usual with our current governance, team and individual scopes of responsibility and reporting structures. As we develop plans for integrating our companies upon closing, we are committed to communicating transparently and continuing to support the wellbeing of our employees.
About our jobs
| 7. | How will the two companies be integrated? Will Blueprint be a separate business unit or division of Sanofi following the closing? |
Until the transaction closes, Blueprint will continue to operate as a separate, independent company, in part due to customary legal requirements. Sanofi has shared that they are impressed with what the Blue Crew has accomplished and excited to build upon our team and welcome us as part of the Sanofi family. We do not yet know how our structure may evolve post-closing. Those plans will be thoughtfully worked through over the next few months before closing. It is common for that level of detail to evolve during this phase of the process and, until closing, no actions are legally permitted to be taken.
During the time between announcement and closing, it’s important that all of us continue to focus on our current priorities on meeting our goals to ensure that we continue to serve patients, customers and partners with the same dedication and integrity that we always have. Leaders from both companies will soon begin to work together to develop integration plans that will guide us once the transaction closes. Throughout this process, we are committed to communicating transparently and continuing to support the wellbeing of our employees.
| 8. | Will my responsibilities or performance expectations change? |
Between now and closing, nothing changes about your role, your responsibilities, performance expectations, or compensation and benefits. We remain an independent company during this period, and that means we will continue to focus on our goals, priorities, and the standards we’ve already set for this year. Your 2025 goals remain the same, and your contributions matter just as much now as they did before the announcement. Looking ahead, once the deal closes, there may be changes but those shifts, if and when they come, will be rolled out thoughtfully and communicated clearly. If you’re unsure about any part of your role, or if your priorities are shifting due to the work we’re doing, please talk to your manager.
| 9. | Is there any work that should stop? |
Until the transaction closes, we will continue to operate as an independent company. This means we will continue to be accountable for operating our business with the same focus, discipline and momentum that got us to this point. Unless you have been specifically informed by your ET member or manager of any changes, you should continue to focus on your day-to-day work. As always, if you have questions or see opportunities for us to operate more effectively or efficiently, please bring them forward to your manager or ET member.
| 10. | Should I seek to engage or work with my counterparts at Sanofi between announcement and closing? |
Until the transaction closes, we will continue to operate as a separate, independent company. Accordingly, no one, except for members of the integration planning team, should coordinate, discuss business or the transaction, or otherwise engage with your counterparts at Sanofi. Remember, this is business as usual. If anyone at Sanofi reaches out to you, please do not respond and talk with your manager. This is important to help us comply with applicable legal requirements between the announcement and closing.
| 11. | Will any roles be eliminated as a result of the transaction between the announcement and closing? When will we know if any roles will be eliminated following closing? |
This transaction is strong recognition of the significant accomplishments our team has delivered, and Sanofi has expressed how energized they are to work alongside members of the Blue Crew to bring life-changing innovations to even more patients around the world. Until the transaction closes, which we anticipate to happen in the third quarter, we will operate business as usual and in the ordinary course.
Over the next few months, integration teams staffed by leaders from both companies will work together to begin integration plans. This learning process will take time to develop a thoughtful plan about how to integrate, where to align, and how to best set us up for long-term success. With a transaction of this size, we would expect some organizational changes to occur over time that could impact some people’s roles. Importantly, we will be transparent and communicate with you regularly throughout the transition process as we work through more detailed integration plans, including any potential impact for members of the Blue Crew.
| 12. | If my role is eliminated after the transaction closes, will I be offered severance benefits? |
We have a Change in Control Severance Pay Plan, or a CIC plan, for each country where we employ Blue Crew members. Each CIC Plan provides that if, within one year after a change in control (referred to in the plans as a “sale event”), an employee's role is eliminated, the employee will be eligible for severance benefits as outlined in the plan. These plans are available for your review on the dedicated transaction page we have created on BLINK. Under the CIC Plans, two criteria must be met to trigger severance benefits eligibility. First, the transaction closes and, second, an employee’s role is eliminated within one year following the closing. Please note that any employee eligible to receive the severance benefits described in the CIC Plans will also be eligible for a career transition assistance benefit.
| 13. | Will I be eligible to receive an annual bonus or field team incentive compensation for 2025? |
If you continue employment through the date that our 2025 annual bonus or final incentive compensation is paid out, you will be eligible to receive a payout based on actual performance. If your role is eliminated following the close but prior to that payment date, then in addition to all of the severance benefits offered in the CIC plan applicable to you, you will receive your 2025 target annual bonus/field team incentive compensation (less any incentive compensation you have already received), pro-rated for the time you are employed in 2025.
| 14. | What will happen to our Cambridge and Zug office locations? Will any employees have to relocate? |
No decisions about potential changes to our office locations post-closing have been made at this early point. Over the next few months, we will help Sanofi better understand our business, ways of working and physical space so that thoughtful decisions can be made. In the meantime, we will operate business as usual at all our existing locations. If office locations should change in the future, we will let you know as soon as possible.
| 15. | What happens to recruiting efforts between the announcement and closing? Are existing offers to candidates still valid? What happens to positions in active interviews? What happens to our contractor workforce? |
We will continue to actively recruit for a defined set of open roles that are essential to supporting this phase of the launch and maintaining the momentum we have built. The Talent Acquisition team will be providing direct updates to hiring managers and candidates related to these prioritized roles in the coming days.
We generally expect that any additional open roles or new backfill requests will be paused through the time between announcement and close and will be part of ongoing integration discussions once they kick-off. We encourage you to talk with your ET member or HR business partner about specific roles or new openings if you have questions or are unsure how to proceed.
The existing offers to candidates who have accepted but not yet started are not impacted, and we look forward to welcoming these new hires. Any offers that are outstanding and pending a candidate decision are also not impacted. In addition, we will continue our relationships with our contractor workforce to the extent they remain aligned with active business priorities during this time between the announcement and close.
Our Talent Acquisition team will be in close contact with all candidates that are active across roles as well as our extended workforce partners to ensure supportive candidate and workforce engagement and clarity. In addition to the Talent Acquisition team, hiring managers can also reach out to your ET member or HR business partner for additional support if needed.
About compensation and benefits
| 16. | Will our compensation and benefits change as a result of the transaction (medical, 401K, leave, etc.)? |
Between the announcement and closing, we will remain two separate entities, and there will generally be no change to your compensation and benefits during this time as a result of our announcement of the transaction. Sanofi has committed that for as long as employees remain employed and for at least a one-year period after closing, it will maintain for each employee (a) at least the same base salary and target cash bonus / cash incentive compensation, and (b) health, welfare and retirement (e.g., 401(k)) benefits that, taken together, are either (i) at least as favorable as those types of Blueprint’s benefits, or (ii) the same benefits provided to similarly situated employees of Sanofi or its subsidiaries. Following closing, we will begin an integration period where Blueprint employees will have an opportunity to learn more about Sanofi’s offerings. Throughout this process of integration, we will communicate transparently, sharing new information as we have it.
| 17. | What will happen to my Blueprint equity? |
Employee equity includes shares acquired through Employee Stock Purchase Program purchases, purchases on the stock market, and settlement of vested restricted stock units (RSUs), as well as stock option and RSU equity awards.
After closing, any shares that are owned and equity awards that are vested will be exchangeable for a cash payment. In addition, any equity awards that were granted before 2025 and are unvested will be fully accelerated and converted to cash. For awards granted in 2025, any vested portions will be exchangeable for cash at close, plus 50% of the unvested portions will vest and be converted to cash at close. The remaining 50% of the unvested award will vest upon the earliest of six months after closing or elimination of the employee’s role as described in the applicable CIC Plan.
Shares, including RSUs will be paid out at $129 per share. Stock options will be paid out at the difference between $129 per option and its associated exercise price. Each share that is converted to cash will also have a CVR, which is a promise of future payment if certain milestones are met (see question #2). Cash payments will be paid on the first payroll after closing or vesting, as applicable.
| 18. | Why will the payout of 50% of my unvested 2025 equity award be delayed until 6 months after closing instead of paying out at closing? |
2025 equity award grants generally have a 4-year vesting schedule completing in 2029. While 50% of these unvested awards will not pay out at closing, Blueprint and Sanofi have agreed to accelerate and pay out such unvested equity awards before 2029 to recognize the significant contributions of Blueprint employees in building the company and advancing our business. In addition, Sanofi has expressed how energized they are to work alongside members of the Blue Crew, and the payout structure supports Sanofi’s vision for continuity during integration and ultimately the long-term success of our business.
| 19. | How will the announcement of the transaction impact our Employee Stock Purchase Program (ESPP)? |
With respect to the offering period under the ESPP that ended on May 31, 2025, your options under the ESPP will be exercised to purchase shares of common stock. Shares of common stock acquired under the ESPP will be treated like other shares of common stock in the transaction. There will be no further offerings under the ESPP, and any remaining cash contributions to the ESPP that were not used in connection with the final option exercise will be returned to you.
| 20. | Will we still complete the mid-year 2025 performance review cycle, including promotions? |
The mid-year performance conversations between employees and managers should be completed as planned. These discussions are a core part of how we support growth, recognize contributions, and stay aligned. Given the timing of the acquisition and the transition ahead, we will not conclude our standard mid-year promotion process as we typically would. We know that recognition and career progression matter deeply and this doesn’t mean that performance isn’t being recognized — it means we’re aligning the timing of formal talent decisions with future talent discussions post-close.
Getting more information
| 21. | Who can I talk to if I have questions? |
We are committed to supporting the Blue Crew and addressing questions as best we can with information and resources. We also know that some members of the Blue Crew may have special considerations, whether it may be an upcoming leave or a pending visa/work permit, that require special support and care. If you have questions or need more clarity, please don’t hesitate to reach out to your ET member, manager, or HR business partner — we’re here to support you and are committed to transparency every step of the way. In addition, you can find a dedicated page on Blink with a repository of information, which we’ll continue to update to keep you informed.
Additional Information about the Proposed Transaction and Where to Find It
The tender offer referred to in this communication has not yet commenced. This communication is for informational purposes only and is neither an offer to purchase nor a solicitation of an offer to sell shares, nor is it a substitute for the tender offer materials that Sanofi and its acquisition subsidiary will file with the U.S. Securities and Exchange Commission (the “SEC”) upon commencement of the tender offer. At the time the tender offer is commenced, Sanofi and its acquisition subsidiary will cause to be filed a tender offer statement on Schedule TO with the SEC, and Blueprint Medicines Corporation (the “Company”) will file a solicitation/recommendation statement on Schedule 14D-9 with respect to the tender offer. THE TENDER OFFER STATEMENT (INCLUDING AN OFFER TO PURCHASE, A RELATED LETTER OF TRANSMITTAL AND OTHER OFFER DOCUMENTS) AND THE SOLICITATION/RECOMMENDATION STATEMENT WILL CONTAIN IMPORTANT INFORMATION THAT SHOULD BE READ CAREFULLY AND CONSIDERED BY THE COMPANY’S STOCKHOLDERS BEFORE ANY DECISION IS MADE WITH RESPECT TO THE TENDER OFFER. Both the tender offer statement and the solicitation/recommendation statement will be mailed to the Company’s stockholders free of charge. A free copy of the tender offer statement and the solicitation/recommendation statement will also be made available to all stockholders of the Company by accessing www.blueprintmedicines.com or by contacting the Company’s Investor Relations contact at ir@blueprintmedicines.com. In addition, the tender offer statement and the solicitation/recommendation statement (and all other documents filed with the SEC) will be available at no charge on the SEC’s website: www.sec.gov, upon filing with the SEC.
THE COMPANY’S STOCKHOLDERS ARE ADVISED TO READ THE SCHEDULE TO AND THE SCHEDULE 14D-9, AS EACH MAY BE AMENDED OR SUPPLEMENTED FROM TIME TO TIME, AND ANY OTHER RELEVANT DOCUMENTS FILED WITH THE SEC WHEN THEY BECOME AVAILABLE BEFORE THEY MAKE ANY DECISION WITH RESPECT TO THE TENDER OFFER, BECAUSE THEY WILL CONTAIN IMPORTANT INFORMATION ABOUT THE PROPOSED TRANSACTION AND THE PARTIES THERETO.
Cautionary Statement Regarding Forward-Looking Statements
This communication contains forward-looking statements regarding, among other things, the proposed acquisition of the Company by Sanofi, the expected timetable for completing the transaction, and the Company’s future financial or operating performance. The Company generally identifies forward-looking statements by terminology such as “aim,” “may,” “will,” “could,” “would,” “should,” “expect,” “plan,” “anticipate,” “intend,” “believe,” “estimate,” “predict,” “project,” “opportunity,” “contemplate,” “potential,” “continue,” “target” or the negative of these terms or other similar words, although not all forward-looking statements contain such terms. All statements that are not statements of historical facts are, or may be deemed to be, forward-looking statements. These forward-looking statements are only predictions, and such statements are based on current expectations and projections about future events and trends as well as the beliefs and assumptions of management. Forward-looking statements are subject to a number of risks and uncertainties, many of which involve factors or circumstances that are beyond the Company’s control. Actual results could differ materially from those stated or implied in forward-looking statements due to a number of factors, including but not limited to: (i) risks associated with the timing of the closing of the proposed transaction, including the risks that a condition to closing would not be satisfied within the expected timeframe or at all or that the closing of the proposed transaction will not occur; (ii) uncertainties as to how many of the Company’s stockholders will tender their shares in the offer; (iii) the possibility that a governmental entity may prohibit, delay or refuse to grant approval for the consummation of the transaction; (iv) the possibility that competing offers will be made; (v) the occurrence of any event, change or other circumstance that could give rise to the termination of the transaction; (vi) the outcome of any legal proceedings that may be instituted against the parties and others related to the merger agreement; (vii) unanticipated difficulties or expenditures relating to the proposed transaction, the response of business partners and competitors to the announcement of the proposed transaction, and/or potential difficulties in employee retention as a result of the announcement and pendency of the proposed transaction; (viii) risks related to non-achievement of the contingent value right (“CVR”) milestones and that holders of the CVRs will not receive payments in respect of the CVRs; (ix) the risk that the marketing and sale of AYVAKIT/ AYVAKYT or any future approved drugs may be unsuccessful or less successful than anticipated, or that AYVAKIT/ AYVAKYT may not gain market acceptance by physicians, patients, third-party payors and others in the medical community; (x) the risk that the market opportunities for AYVAKIT/ AYVAKYT or the Company’s drug candidates are smaller than the Company estimates or that any approval that the Company obtains may be based on a narrower definition of the patient population that the Company anticipates; (xi) the risk of delay of any current or planned clinical trials or the development of the Company’s current or future drug candidates, including but not limited to BLU-808 and elenestinib; (xii) risks related to the Company’s ability to successfully demonstrate the safety and efficacy of its drug candidates and gain approval of its drug candidates on a timely basis, if at all; (xiii) preclinical and clinical results for the Company’s drug candidates may not support further development of such drug candidates either as monotherapies or in combination with other agents or may impact the anticipated timing of data or regulatory submissions; (xiv) the timing of the initiation of clinical trials and trial cohorts at clinical trial sites and patient enrollment rates may be delayed or slower than anticipated; (xv) actions of regulatory agencies may affect the Company’s approved drugs or its current or future drug candidates, including affecting the initiation, timing and progress of clinical trials, as well as the pricing of its drug candidates; (xvi) risks related to the Company’s ability to obtain, maintain and enforce patent and other intellectual property protection for its products and current or future drug candidates it is developing; (xvii) the success of the Company’s current and future collaborations, financing arrangements, partnerships or licensing and other arrangements; (xviii) risks related to the Company’s liquidity and financial position and the accuracy of its estimates of revenues, expenses, cash burn, and capital requirements; and (xix) those risks detailed in the Company’s most recent Annual Report on Form 10-K and subsequent reports filed with the SEC, as well as other documents that may be filed by the Company from time to time with the SEC. The Company cannot assure you that the events and circumstances reflected in the forward-looking statements will be achieved or occur, and actual results could differ materially from those projected in the forward-looking statements. The forward-looking statements made in this communication relate only to events as of the date on which the statements are made. The Company undertakes no obligation to update any forward-looking statement to reflect events or circumstances after the date on which the statement is made or to reflect the occurrence of unanticipated events.
Exhibit 99.3
X Post
LinkedIn Post
Additional Information about the Proposed Transaction and Where to Find It
The tender offer referred to in this communication has not yet commenced. This communication is for informational purposes only and is neither an offer to purchase nor a solicitation of an offer to sell shares, nor is it a substitute for the tender offer materials that Sanofi and its acquisition subsidiary will file with the U.S. Securities and Exchange Commission (the “SEC”) upon commencement of the tender offer. At the time the tender offer is commenced, Sanofi and its acquisition subsidiary will cause to be filed a tender offer statement on Schedule TO with the SEC, and Blueprint Medicines Corporation (the “Company”) will file a solicitation/recommendation statement on Schedule 14D-9 with respect to the tender offer. THE TENDER OFFER STATEMENT (INCLUDING AN OFFER TO PURCHASE, A RELATED LETTER OF TRANSMITTAL AND OTHER OFFER DOCUMENTS) AND THE SOLICITATION/RECOMMENDATION STATEMENT WILL CONTAIN IMPORTANT INFORMATION THAT SHOULD BE READ CAREFULLY AND CONSIDERED BY THE COMPANY’S STOCKHOLDERS BEFORE ANY DECISION IS MADE WITH RESPECT TO THE TENDER OFFER. Both the tender offer statement and the solicitation/recommendation statement will be mailed to the Company’s stockholders free of charge. A free copy of the tender offer statement and the solicitation/recommendation statement will also be made available to all stockholders of the Company by accessing www.blueprintmedicines.com or by contacting the Company’s Investor Relations contact at ir@blueprintmedicines.com. In addition, the tender offer statement and the solicitation/recommendation statement (and all other documents filed with the SEC) will be available at no charge on the SEC’s website: www.sec.gov, upon filing with the SEC.
THE COMPANY’S STOCKHOLDERS ARE ADVISED TO READ THE SCHEDULE TO AND THE SCHEDULE 14D-9, AS EACH MAY BE AMENDED OR SUPPLEMENTED FROM TIME TO TIME, AND ANY OTHER RELEVANT DOCUMENTS FILED WITH THE SEC WHEN THEY BECOME AVAILABLE BEFORE THEY MAKE ANY DECISION WITH RESPECT TO THE TENDER OFFER, BECAUSE THEY WILL CONTAIN IMPORTANT INFORMATION ABOUT THE PROPOSED TRANSACTION AND THE PARTIES THERETO.
Cautionary Statement Regarding Forward-Looking Statements
This communication contains forward-looking statements regarding, among other things, the proposed acquisition of the Company by Sanofi, the expected timetable for completing the transaction, and the Company’s future financial or operating performance. The Company generally identifies forward-looking statements by terminology such as “aim,” “may,” “will,” “could,” “would,” “should,” “expect,” “plan,” “anticipate,” “intend,” “believe,” “estimate,” “predict,” “project,” “opportunity,” “contemplate,” “potential,” “continue,” “target” or the negative of these terms or other similar words, although not all forward-looking statements contain such terms. All statements that are not statements of historical facts are, or may be deemed to be, forward-looking statements. These forward-looking statements are only predictions, and such statements are based on current expectations and projections about future events and trends as well as the beliefs and assumptions of management. Forward-looking statements are subject to a number of risks and uncertainties, many of which involve factors or circumstances that are beyond the Company’s control. Actual results could differ materially from those stated or implied in forward-looking statements due to a number of factors, including but not limited to: (i) risks associated with the timing of the closing of the proposed transaction, including the risks that a condition to closing would not be satisfied within the expected timeframe or at all or that the closing of the proposed transaction will not occur; (ii) uncertainties as to how many of the Company’s stockholders will tender their shares in the offer; (iii) the possibility that a governmental entity may prohibit, delay or refuse to grant approval for the consummation of the transaction; (iv) the possibility that competing offers will be made; (v) the occurrence of any event, change or other circumstance that could give rise to the termination of the transaction; (vi) the outcome of any legal proceedings that may be instituted against the parties and others related to the merger agreement; (vii) unanticipated difficulties or expenditures relating to the proposed transaction, the response of business partners and competitors to the announcement of the proposed transaction, and/or potential difficulties in employee retention as a result of the announcement and pendency of the proposed transaction; (viii) risks related to non-achievement of the contingent value right (“CVR”) milestones and that holders of the CVRs will not receive payments in respect of the CVRs; (ix) the risk that the marketing and sale of AYVAKIT/ AYVAKYT or any future approved drugs may be unsuccessful or less successful than anticipated, or that AYVAKIT/ AYVAKYT may not gain market acceptance by physicians, patients, third-party payors and others in the medical community; (x) the risk that the market opportunities for AYVAKIT/ AYVAKYT or the Company’s drug candidates are smaller than the Company estimates or that any approval that the Company obtains may be based on a narrower definition of the patient population that the Company anticipates; (xi) the risk of delay of any current or planned clinical trials or the development of the Company’s current or future drug candidates, including but not limited to BLU-808 and elenestinib; (xii) risks related to the Company’s ability to successfully demonstrate the safety and efficacy of its drug candidates and gain approval of its drug candidates on a timely basis, if at all; (xiii) preclinical and clinical results for the Company’s drug candidates may not support further development of such drug candidates either as monotherapies or in combination with other agents or may impact the anticipated timing of data or regulatory submissions; (xiv) the timing of the initiation of clinical trials and trial cohorts at clinical trial sites and patient enrollment rates may be delayed or slower than anticipated; (xv) actions of regulatory agencies may affect the Company’s approved drugs or its current or future drug candidates, including affecting the initiation, timing and progress of clinical trials, as well as the pricing of its drug candidates; (xvi) risks related to the Company’s ability to obtain, maintain and enforce patent and other intellectual property protection for its products and current or future drug candidates it is developing; (xvii) the success of the Company’s current and future collaborations, financing arrangements, partnerships or licensing and other arrangements; (xviii) risks related to the Company’s liquidity and financial position and the accuracy of its estimates of revenues, expenses, cash burn, and capital requirements; and (xix) those risks detailed in the Company’s most recent Annual Report on Form 10-K and subsequent reports filed with the SEC, as well as other documents that may be filed by the Company from time to time with the SEC. The Company cannot assure you that the events and circumstances reflected in the forward-looking statements will be achieved or occur, and actual results could differ materially from those projected in the forward-looking statements. The forward-looking statements made in this communication relate only to events as of the date on which the statements are made. The Company undertakes no obligation to update any forward-looking statement to reflect events or circumstances after the date on which the statement is made or to reflect the occurrence of unanticipated events.
Exhibit 99.4
Good morning,
This morning, Blueprint announced that we have reached an agreement to be acquired by Sanofi for approximately $9.5 billion, or up to $135 per share in total consideration.
Key highlights of the transaction include:
| · | The per share purchase price represents a 33%, 35%, and 49% premium to the company’s current share price, 30– and 60-day volume-weighted average prices as of May 30, 2025, respectively. | |
| · | In addition to $129 per share in cash at closing, Blueprint shareholders will also receive one non-tradeable Contingent Value Right (CVR) for each Blueprint share held. The CVRs represent the right to receive two contingent payments of $2 and $4 per share in cash related to the achievement of certain clinical development and regulatory milestones for BLU-808. This represents additional potential upside value for a pipeline that remains in development. | |
| · | The transaction is expected to close in the third quarter of 2025, subject to customary closing conditions and approvals. |
This is an important milestone in our journey as a company, and we believe there is no better strategic fit than with Sanofi. Blueprint’s pipeline of innovative commercial- and development-stage therapies for mast cell diseases aligns seamlessly with Sanofi global leadership in rare diseases and immunology. With exceptional strength and global infrastructure in these areas spanning discovery to commercialization, Sanofi will enable us to accelerate our mission by expanding the reach and impact of our portfolio to many more patients globally. We are also pleased that our agreement with Sanofi will deliver significant, immediate, and certain value to our stockholders, and with the potential to receive additional value in the future through the CVRs.
You can read more about the transaction in the attached press release. [In addition, Sanofi plans to host a Zoom call for investors and analysts at [8:30 am CEST / 2:30 am ET]. The access information for the can be found in Sanofi’s press release and is also pasted below for convenience.
https://sanofi.zoom.us/j/97991465119?pwd=KHb1Zwhgh8e8UrgipSpUEt9PD1VizA.1
Webinar ID: 979 9146 5119
Passcode: 801394]
As always, thank you for your support of Blueprint Medicines,
Jenna
Jenna Cohen
VP, Investor Relations and Global Business Communications
Blueprint Medicines
Additional Information about the Proposed Transaction and Where to Find It
The tender offer referred to in this communication has not yet commenced. This communication is for informational purposes only and is neither an offer to purchase nor a solicitation of an offer to sell shares, nor is it a substitute for the tender offer materials that Sanofi and its acquisition subsidiary will file with the U.S. Securities and Exchange Commission (the “SEC”) upon commencement of the tender offer. At the time the tender offer is commenced, Sanofi and its acquisition subsidiary will cause to be filed a tender offer statement on Schedule TO with the SEC, and Blueprint Medicines Corporation (the “Company”) will file a solicitation/recommendation statement on Schedule 14D-9 with respect to the tender offer. THE TENDER OFFER STATEMENT (INCLUDING AN OFFER TO PURCHASE, A RELATED LETTER OF TRANSMITTAL AND OTHER OFFER DOCUMENTS) AND THE SOLICITATION/RECOMMENDATION STATEMENT WILL CONTAIN IMPORTANT INFORMATION THAT SHOULD BE READ CAREFULLY AND CONSIDERED BY THE COMPANY’S STOCKHOLDERS BEFORE ANY DECISION IS MADE WITH RESPECT TO THE TENDER OFFER. Both the tender offer statement and the solicitation/recommendation statement will be mailed to the Company’s stockholders free of charge. A free copy of the tender offer statement and the solicitation/recommendation statement will also be made available to all stockholders of the Company by accessing www.blueprintmedicines.com or by contacting the Company’s Investor Relations contact at ir@blueprintmedicines.com. In addition, the tender offer statement and the solicitation/recommendation statement (and all other documents filed with the SEC) will be available at no charge on the SEC’s website: www.sec.gov, upon filing with the SEC.
THE COMPANY’S STOCKHOLDERS ARE ADVISED TO READ THE SCHEDULE TO AND THE SCHEDULE 14D-9, AS EACH MAY BE AMENDED OR SUPPLEMENTED FROM TIME TO TIME, AND ANY OTHER RELEVANT DOCUMENTS FILED WITH THE SEC WHEN THEY BECOME AVAILABLE BEFORE THEY MAKE ANY DECISION WITH RESPECT TO THE TENDER OFFER, BECAUSE THEY WILL CONTAIN IMPORTANT INFORMATION ABOUT THE PROPOSED TRANSACTION AND THE PARTIES THERETO.
Cautionary Statement Regarding Forward-Looking Statements
This communication contains forward-looking statements regarding, among other things, the proposed acquisition of the Company by Sanofi, the expected timetable for completing the transaction, and the Company’s future financial or operating performance. The Company generally identifies forward-looking statements by terminology such as “aim,” “may,” “will,” “could,” “would,” “should,” “expect,” “plan,” “anticipate,” “intend,” “believe,” “estimate,” “predict,” “project,” “opportunity,” “contemplate,” “potential,” “continue,” “target” or the negative of these terms or other similar words, although not all forward-looking statements contain such terms. All statements that are not statements of historical facts are, or may be deemed to be, forward-looking statements. These forward-looking statements are only predictions, and such statements are based on current expectations and projections about future events and trends as well as the beliefs and assumptions of management. Forward-looking statements are subject to a number of risks and uncertainties, many of which involve factors or circumstances that are beyond the Company’s control. Actual results could differ materially from those stated or implied in forward-looking statements due to a number of factors, including but not limited to: (i) risks associated with the timing of the closing of the proposed transaction, including the risks that a condition to closing would not be satisfied within the expected timeframe or at all or that the closing of the proposed transaction will not occur; (ii) uncertainties as to how many of the Company’s stockholders will tender their shares in the offer; (iii) the possibility that a governmental entity may prohibit, delay or refuse to grant approval for the consummation of the transaction; (iv) the possibility that competing offers will be made; (v) the occurrence of any event, change or other circumstance that could give rise to the termination of the transaction; (vi) the outcome of any legal proceedings that may be instituted against the parties and others related to the merger agreement; (vii) unanticipated difficulties or expenditures relating to the proposed transaction, the response of business partners and competitors to the announcement of the proposed transaction, and/or potential difficulties in employee retention as a result of the announcement and pendency of the proposed transaction; (viii) risks related to non-achievement of the contingent value right (“CVR”) milestones and that holders of the CVRs will not receive payments in respect of the CVRs; (ix) the risk that the marketing and sale of AYVAKIT/ AYVAKYT or any future approved drugs may be unsuccessful or less successful than anticipated, or that AYVAKIT/ AYVAKYT may not gain market acceptance by physicians, patients, third-party payors and others in the medical community; (x) the risk that the market opportunities for AYVAKIT/ AYVAKYT or the Company’s drug candidates are smaller than the Company estimates or that any approval that the Company obtains may be based on a narrower definition of the patient population that the Company anticipates; (xi) the risk of delay of any current or planned clinical trials or the development of the Company’s current or future drug candidates, including but not limited to BLU-808 and elenestinib; (xii) risks related to the Company’s ability to successfully demonstrate the safety and efficacy of its drug candidates and gain approval of its drug candidates on a timely basis, if at all; (xiii) preclinical and clinical results for the Company’s drug candidates may not support further development of such drug candidates either as monotherapies or in combination with other agents or may impact the anticipated timing of data or regulatory submissions; (xiv) the timing of the initiation of clinical trials and trial cohorts at clinical trial sites and patient enrollment rates may be delayed or slower than anticipated; (xv) actions of regulatory agencies may affect the Company’s approved drugs or its current or future drug candidates, including affecting the initiation, timing and progress of clinical trials, as well as the pricing of its drug candidates; (xvi) risks related to the Company’s ability to obtain, maintain and enforce patent and other intellectual property protection for its products and current or future drug candidates it is developing; (xvii) the success of the Company’s current and future collaborations, financing arrangements, partnerships or licensing and other arrangements; (xviii) risks related to the Company’s liquidity and financial position and the accuracy of its estimates of revenues, expenses, cash burn, and capital requirements; and (xix) those risks detailed in the Company’s most recent Annual Report on Form 10-K and subsequent reports filed with the SEC, as well as other documents that may be filed by the Company from time to time with the SEC. The Company cannot assure you that the events and circumstances reflected in the forward-looking statements will be achieved or occur, and actual results could differ materially from those projected in the forward-looking statements. The forward-looking statements made in this communication relate only to events as of the date on which the statements are made. The Company undertakes no obligation to update any forward-looking statement to reflect events or circumstances after the date on which the statement is made or to reflect the occurrence of unanticipated events.
Exhibit 99.5

Blueprint Medicines + Sanofi Overview for the Blue Crew June 2, 2025

Blueprint Medicines + Sanofi: a strong strategic fit 2 Well - established global leadership in rare disease and immunology & inflammation Significant resources, infrastructure & capability, and global reach Similar mission and culture centering science and patient impact Leading SM franchise at the intersection of rare disease and immunology & inflammation Mast cell expertise and pipeline at the forefront of innovation in immunology & inflammation Culture of excellence and proven track record of success in delivering innovation to patients Accelerates Blueprint’s mission to deliver life - changing medicines to more patients globally , and creates a cornerstone for Sanofi’s vision of global leadership in immunology

Blueprint Medicines + Sanofi: a strong strategic fit 3 Our portfolio of mast cell targeted therapies , including AYVAKIT, elenestinib and BLU - 808, fit seamlessly with Sanofi’s global leadership in rare disease and immunology & inflammation Opportunity to maximize the pipeline - in - a - product potential of BLU - 808 by leveraging Sanofi’s resources and strategic development capability in immunology & inflammation The Boards of Directors and Executive Teams of both companies unanimously support the transaction based on the value it immediately recognizes and can create in the future for patients and shareholders Sanofi aims to further build around AYVAKIT, our pipeline and people as a cornerstone for their vision of global leadership in immunology

4 Welcoming Brian Foard Executive Vice President Head of Specialty Care, Sanofi

What happens next? 5 We will operate independently in our current structure and governance. Your responsibilities, compensation and benefits will generally continue in the normal course. Blueprint and Sanofi will work together to develop an integration plan. NOW UNTIL CLOSING UPON CLOSING (ESTIMATED IN Q3 2025) Blueprint becomes part of Sanofi and integration begins.
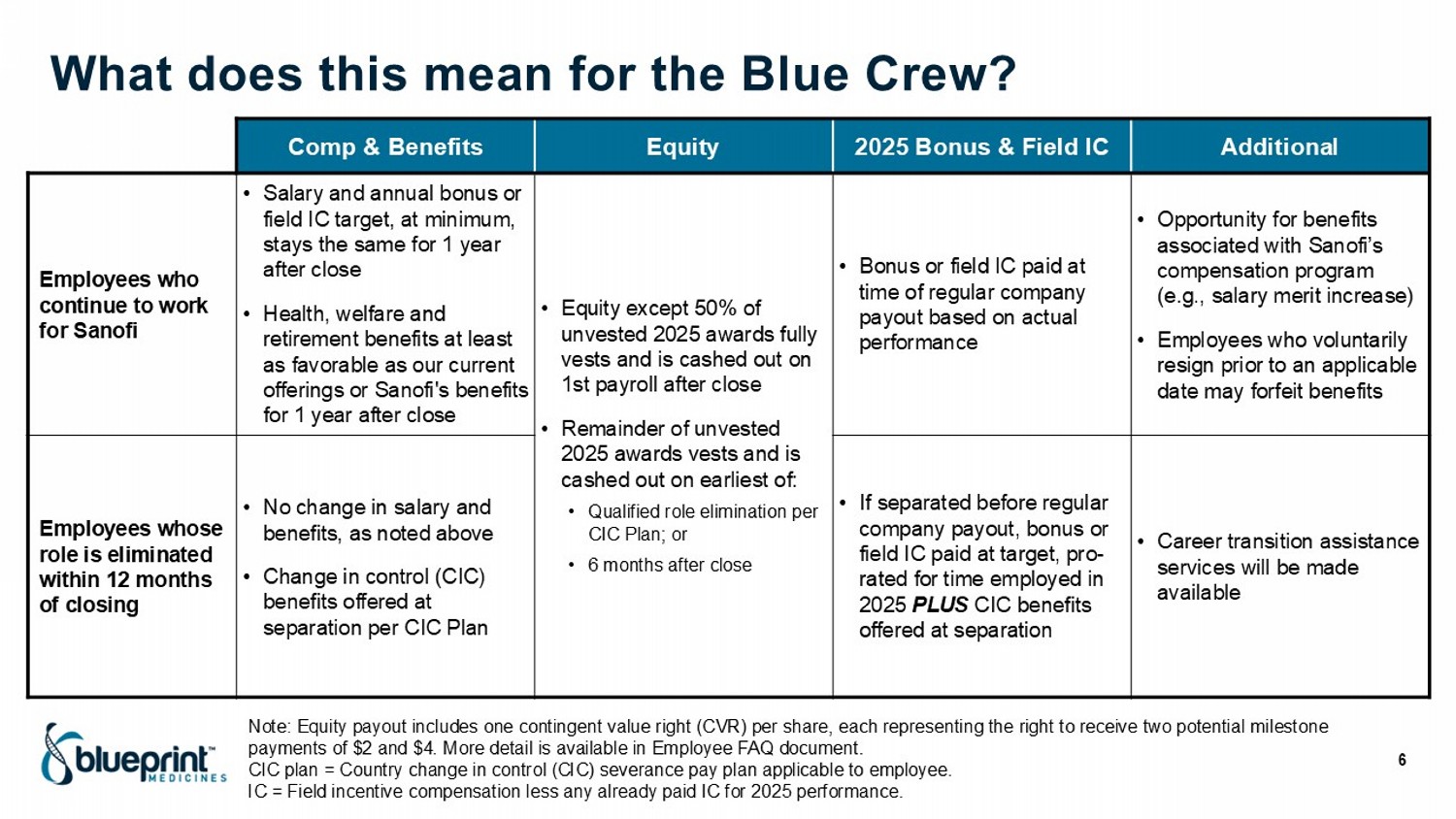
What does this mean for the Blue Crew? 6 Additional 2025 Bonus & Field IC Equity Comp & Benefits • Opportunity for benefits associated with Sanofi’s compensation program (e.g., salary merit increase) • Employees who voluntarily resign prior to an applicable date may forfeit benefits • Bonus or field IC paid at time of regular company payout based on actual performance • Equity except 50% of unvested 2025 awards fully vests and is cashed out on 1st payroll after close • Remainder of unvested 2025 awards vests and is cashed out on earliest of: • Qualified role elimination per CIC Plan; or • 6 months after close • Salary and annual bonus or field IC target, at minimum, stays the same for 1 year after close • Health, welfare and retirement benefits at least as favorable as our current offerings or Sanofi's benefits for 1 year after close Employees who continue to work for Sanofi • Career transition assistance services will be made available • If separated before regular company payout, bonus or field IC paid at target, pro - rated for time employed in 2025 PLUS CIC benefits offered at separation • No change in salary and benefits, as noted above • Change in control (CIC) benefits offered at separation per CIC Plan Employees whose role is eliminated within 12 months of closing Note: Equity payout includes one contingent value right (CVR) per share, each representing the right to receive two potential mi lestone payments of $2 and $4. More detail is available in Employee FAQ document. CIC plan = Country change in control (CIC) severance pay plan applicable to employee. IC = Field incentive compensation less any already paid IC for 2025 performance.

Speaking with one voice: communicating prior to closing 7 Media and Investor Inquiries Please do not respond and refer all inquiries to Jim Baker, SVP Corporate Affairs ( media@blueprintmedicines.com ) or Jenna Cohen, VP Investor Relations & Global Business Communications ( ir@blueprintmedicines.com ) When speaking with healthcare professionals, patients, patient advocacy groups, and other partners, reinforce continuity and an expectation of continued quality engagement from the Blueprint team. You should… x Continue to operate as you normally would in support of patients and customers x Share appropriate information reactively, referencing the External Stakeholder Talking Points and Template Email documents available on Blink x Align proactive communication to priority stakeholders with your ET member or function leader before sending x Direct inquiries to the press release for further detail You may not… ˣ Respond to media, press, or investor inquiries (see guidance below) ˣ Coordinate, discuss business or the transaction, or otherwise engage with your counterparts at Sanofi, unless cleared first with your supervisor and counsel

We understand that you may still have questions. A dedicated page is available on Blink with information and resources, including: x Press release announcing the transaction x Employee FAQs x Approved messages to respond to external inquiries, including customers x Today’s company meeting slides x Copies of our Change in Control plans We are committed to sharing additional information as it becomes available, and communicating transparently and with clarity. If you have questions, talk to your ET member, manager or HR business partner or send an email to BlueQ@blueprintmedicines.com 8

9 Ask a question Mike Landsittel Debbie Bumpus Fouad Namouni Christy Rossi Kate Haviland CFO CPO President, R&D COO CEO

Additional Information about the Proposed Transaction and Where to Find It 10 The tender offer referred to in this communication has not yet commenced . This communication is for informational purposes only and is neither an offer to purchase nor a solicitation of an offer to sell shares, nor is it a substitute for the tender offer materials that Sanofi and its acquisition subsidiary will file with the U . S . Securities and Exchange Commission (the “SEC”) upon commencement of the tender offer . At the time the tender offer is commenced, Sanofi and its acquisition subsidiary will cause to be filed a tender offer statement on Schedule TO with the SEC, and Blueprint Medicines Corporation (the “Company”) will file a solicitation/recommendation statement on Schedule 14 D - 9 with respect to the tender offer . THE TENDER OFFER STATEMENT (INCLUDING AN OFFER TO PURCHASE, A RELATED LETTER OF TRANSMITTAL AND OTHER OFFER DOCUMENTS) AND THE SOLICITATION/RECOMMENDATION STATEMENT WILL CONTAIN IMPORTANT INFORMATION THAT SHOULD BE READ CAREFULLY AND CONSIDERED BY THE COMPANY’S STOCKHOLDERS BEFORE ANY DECISION IS MADE WITH RESPECT TO THE TENDER OFFER . Both the tender offer statement and the solicitation/recommendation statement will be mailed to the Company’s stockholders free of charge . A free copy of the tender offer statement and the solicitation/recommendation statement will also be made available to all stockholders of the Company by accessing www . blueprintmedicines . com or by contacting the Company’s Investor Relations contact at ir@blueprintmedicines . com . In addition, the tender offer statement and the solicitation/recommendation statement (and all other documents filed with the SEC) will be available at no charge on the SEC’s website : www . sec . gov, upon filing with the SEC . THE COMPANY’S STOCKHOLDERS ARE ADVISED TO READ THE SCHEDULE TO AND THE SCHEDULE 14 D - 9 , AS EACH MAY BE AMENDED OR SUPPLEMENTED FROM TIME TO TIME, AND ANY OTHER RELEVANT DOCUMENTS FILED WITH THE SEC WHEN THEY BECOME AVAILABLE BEFORE THEY MAKE ANY DECISION WITH RESPECT TO THE TENDER OFFER, BECAUSE THEY WILL CONTAIN IMPORTANT INFORMATION ABOUT THE PROPOSED TRANSACTION AND THE PARTIES THERETO .

Cautionary Statement Regarding Forward - Looking Statements 11 This communication contains forward - looking statements regarding, among other things, the proposed acquisition of the Company by Sanofi, the expected timetable for completing the transaction, and the Company’s future financial or operating performance . The Company generally identifies forward - looking statements by terminology such as “aim,” “may,” “will,” “could,” “would,” “should,” “expect,” “plan,” “anticipate,” “intend,” “believe,” “estimate,” “predict,” “project,” “opportunity,” “contemplate,” “potential,” “continue,” “target” or the negative of these terms or other similar words, although not all forward - looking statements contain such terms . All statements that are not statements of historical facts are, or may be deemed to be, forward - looking statements . These forward - looking statements are only predictions, and such statements are based on current expectations and projections about future events and trends as well as the beliefs and assumptions of management . Forward - looking statements are subject to a number of risks and uncertainties, many of which involve factors or circumstances that are beyond the Company’s control . Actual results could differ materially from those stated or implied in forward - looking statements due to a number of factors, including but not limited to : (i) risks associated with the timing of the closing of the proposed transaction, including the risks that a condition to closing would not be satisfied within the expected timeframe or at all or that the closing of the proposed transaction will not occur ; (ii) uncertainties as to how many of the Company’s stockholders will tender their shares in the offer ; (iii) the possibility that a governmental entity may prohibit, delay or refuse to grant approval for the consummation of the transaction ; (iv) the possibility that competing offers will be made ; (v) the occurrence of any event, change or other circumstance that could give rise to the termination of the transaction ; (vi) the outcome of any legal proceedings that may be instituted against the parties and others related to the merger agreement ; (vii) unanticipated difficulties or expenditures relating to the proposed transaction, the response of business partners and competitors to the announcement of the proposed transaction, and/or potential difficulties in employee retention as a result of the announcement and pendency of the proposed transaction ; (viii) risks related to non - achievement of the contingent value right (“CVR”) milestones and that holders of the CVRs will not receive payments in respect of the CVRs ; (ix) the risk that the marketing and sale of AYVAKIT / AYVAKYT or any future approved drugs may be unsuccessful or less successful than anticipated, or that AYVAKIT / AYVAKYT may not gain market acceptance by physicians, patients, third - party payors and others in the medical community ; (x) the risk that the market opportunities for AYVAKIT / AYVAKYT or the Company’s drug candidates are smaller than the Company estimates or that any approval that the Company obtains may be based on a narrower definition of the patient population that the Company anticipates ; (xi) the risk of delay of any current or planned clinical trials or the development of the Company’s current or future drug candidates, including but not limited to BLU - 808 and elenestinib ; (xii) risks related to the Company’s ability to successfully demonstrate the safety and efficacy of its drug candidates and gain approval of its drug candidates on a timely basis, if at all ; (xiii) preclinical and clinical results for the Company’s drug candidates may not support further development of such drug candidates either as monotherapies or in combination with other agents or may impact the anticipated timing of data or regulatory submissions ; (xiv) the timing of the initiation of clinical trials and trial cohorts at clinical trial sites and patient enrollment rates may be delayed or slower than anticipated ; (xv) actions of regulatory agencies may affect the Company’s approved drugs or its current or future drug candidates, including affecting the initiation, timing and progress of clinical trials, as well as the pricing of its drug candidates ; (xvi) risks related to the Company’s ability to obtain, maintain and enforce patent and other intellectual property protection for its products and current or future drug candidates it is developing ; (xvii) the success of the Company’s current and future collaborations, financing arrangements, partnerships or licensing and other arrangements ; (xviii) risks related to the Company’s liquidity and financial position and the accuracy of its estimates of revenues, expenses, cash burn, and capital requirements ; and (xix) those risks detailed in the Company’s most recent Annual Report on Form 10 - K and subsequent reports filed with the SEC, as well as other documents that may be filed by the Company from time to time with the SEC . The Company cannot assure you that the events and circumstances reflected in the forward - looking statements will be achieved or occur, and actual results could differ materially from those projected in the forward - looking statements . The forward - looking statements made in this communication relate only to events as of the date on which the statements are made . The Company undertakes no obligation to update any forward - looking statement to reflect events or circumstances after the date on which the statement is made or to reflect the occurrence of unanticipated events .
