UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
SCHEDULE TO
Tender Offer Statement under Section 14(d)(1) or 13(e)(1)
of the Securities Exchange Act of 1934
Blueprint Medicines Corporation
(Name of Subject Company)
ROTHKO MERGER SUB, INC.
AVENTIS INC.
SANOFI
(Names of Filing Persons — Offerors)
Common Stock, Par Value $0.001 Per Share
(Title of Class of Securities)
09627Y109
(Cusip Number of Class of Securities)
Roy Papatheodorou
Executive Vice President, General Counsel, Head of Legal Ethics & Business Integrity
Sanofi
46, avenue de la Grande Armée, 75017
Paris, France
Telephone: 011 + 33 1 53 77 40 00
(Name, Address and Telephone Number of Person Authorized to Receive Notices and Communications on Behalf of Filing Persons)
Copies to:
Michael J. Aiello, Esq.
Sachin Kohli, Esq.
Weil, Gotshal & Manges LLP
767 Fifth Avenue New York, New York 10153
(212) 310-8000
CALCULATION OF FILING FEE
| Transaction Valuation* | Amount of Filing Fee* | |
| N/A | N/A |
| * | A filing fee is not required in connection with this filing as it relates solely to preliminary communications made before the commencement of a tender offer. |
| ☐ | Check box if any part of the fee is offset as provided by Rule 0-11(a)(2) and identify the filing with which the offsetting fee was previously paid. Identify the previous filing by registration statement number, or the Form or Schedule and the date of its filing. |
| Amount Previously Paid: Not applicable. | Filing Party: Not applicable. | |||
| Form or Registration No.: Not applicable | Date Filed: Not applicable. |
| ☒ | Check the box if the filing relates solely to preliminary communications made before the commencement of a tender offer. |
Check the appropriate boxes below to designate any transactions to which the statement relates:
| ☒ | third-party tender offer subject to Rule 14d-1. |
| ☐ | issuer tender offer subject to Rule 13e-4. |
| ☐ | going-private transaction subject to Rule 13e-3. |
| ☐ | amendment to Schedule 13D under Rule 13d-2. |
Check the following box if the filing is a final amendment reporting the results of the tender offer. ☐
This Schedule TO-C consists of the following documents related to the proposed acquisition of Blueprint Medicines Corporation:
| 1. | Post on employee portal to certain Sanofi employees from Brian Foard, dated June 2, 2025, a copy of which is attached hereto as Exhibit 99.1 and incorporated herein by reference. |
| 2. | Post on employee portal to Sanofi research and development organization from Houman Ashrafian, dated June 2, 2025, a copy of which is attached hereto as Exhibit 99.2 and incorporated herein by reference. |
| 3. | Social media posts on Sanofi’s LinkedIn and X accounts, dated June 2, 2025, a copy of which is attached hereto as Exhibit 99.3 and incorporated herein by reference. |
| 4. | Slide deck presented on investor relations call, dated June 2, 2025, a copy of which is attached hereto as Exhibit 99.4 and incorporated herein by reference. |
| 5. | Post on employee portal to Sanofi employees, dated June 2, 2025, a copy of which is attached hereto as Exhibit 99.5 and incorporated herein by reference. |
| 6. | Investor Relations call transcript, dated June 2, 2025, a copy of which is attached hereto as Exhibit 99.6 and incorporated herein by reference. |
The items listed above were first used or made available on June 2, 2025.
Additional Information for US Shareholders and Where to Find It
The tender offer for the outstanding shares of Blueprint Medicines Corporation common stock (“Blueprint”) referenced in this communication has not yet commenced. This communication is for informational purposes only and is neither an offer to purchase nor a solicitation of an offer to sell shares of Blueprint, nor is it a substitute for the tender offer materials that Sanofi and its acquisition subsidiary will file with the U.S. Securities and Exchange Commission (the “SEC”) upon commencement of the tender offer. At the time the tender offer is commenced, Sanofi and its acquisition subsidiary will file tender offer materials on Schedule TO, and Blueprint will file a Solicitation/Recommendation Statement on Schedule 14D-9 with the SEC with respect to the tender offer. The tender offer materials (including an Offer to Purchase, a related Letter of Transmittal and certain other tender offer documents) and the Solicitation/Recommendation Statement will contain important information. HOLDERS OF SHARES OF BLUEPRINT ARE URGED TO READ THESE DOCUMENTS WHEN THEY BECOME AVAILABLE BECAUSE THEY WILL CONTAIN IMPORTANT INFORMATION THAT BLUEPRINT STOCKHOLDERS SHOULD CONSIDER BEFORE MAKING ANY DECISION REGARDING TENDERING THEIR SHARES. The Offer to Purchase, the related Letter of Transmittal and certain other tender offer documents, as well as the Solicitation/Recommendation Statement, will be made available to all holders of shares of Blueprint at no expense to them. The tender offer materials and the Solicitation/Recommendation Statement will be made available for free at the SEC’s web site at www.sec.gov. Additional copies may be obtained for free by contacting Sanofi’s Investor Relations Team at investor.relations@sanofi.com or on Sanofi’s website at https://www.sanofi.com/en/investors.
In addition to the Offer to Purchase, the related Letter of Transmittal and certain other tender offer documents, as well as the Solicitation/Recommendation Statement, Sanofi files annual and special reports and other information with the SEC and Blueprint files annual, quarterly and special reports and other information with the SEC. You may read and copy any reports or other information filed by Sanofi and Blueprint at the SEC public reference room at 100 F. Street, N.E., Washington D.C. 20549. Please call the Commission at 1-800-SEC-0330 for further information on the public reference room. Sanofi’s and Blueprint’s filings with the SEC are also available to the public from commercial document-retrieval services and at the website maintained by the SEC at www.sec.gov.
Cautionary Statement Regarding Forward-Looking Statements
This communication contains forward-looking statements that are subject to risks, uncertainties and other factors that could cause actual results to differ materially from those implied by the forward-looking statements. Forward-looking statements are statements that are not historical facts and may include projections and estimates and their underlying assumptions, statements regarding plans, objectives, intentions and expectations with respect to future financial results, events, operations, services, product development and potential, and statements regarding future performance. Forward-looking statements are generally identified by the words “expects”, “anticipates”, “believes”, “intends”, “estimates”, “plans”, “will be” and similar expressions. Although Sanofi’s management believes that the expectations reflected in such forward-looking statements are reasonable, investors are cautioned that forward-looking information and statements are subject to various risks and uncertainties, many of which are difficult to predict and generally beyond the control of Sanofi, that could cause actual results and developments to differ materially from those expressed in, or implied or projected by, the forward-looking information and statements. These risks and uncertainties include among other things, unexpected regulatory actions or delays, or government regulation generally, that could affect the availability or commercial potential of the product, or the fact that the product may not be commercially successful, and risks related to Sanofi’s and Blueprint’s ability to complete the acquisition on the proposed terms or on the proposed timeline or at all, including the receipt of required regulatory approvals, the risk that the conditions to the closing of the transaction may not be satisfied, the possibility that competing offers will be made, the risks that the milestones related to the contingent value right will not be achieved, the risk of securityholder litigation relating to the proposed acquisition, including resulting expense or delays, other risks associated with executing business combination transactions, such as the risk that the businesses will not be integrated successfully, that such integration may be more difficult, time-consuming or costly than expected or that the expected benefits of the acquisition will not be realized, risks related to future opportunities and plans for the combined company, including uncertainty of the expected financial performance and results of the combined company following completion of the proposed acquisition, disruption from the proposed acquisition making it more difficult to conduct business as usual or to maintain relationships with customers, employees, manufacturers, suppliers or patient groups, and the possibility that, if the combined company does not achieve the perceived benefits of the proposed acquisition as rapidly or to the extent anticipated by financial analysts or investors, the market price of Sanofi’s shares could decline, as well as other risks related to Sanofi’s and Blueprint’s respective businesses, including the ability to grow sales and revenues from existing products and to develop, commercialize or market new products, competition, including potential generic competition, the uncertainties inherent in research and development, including future clinical data and analysis, regulatory obligations and oversight by regulatory authorities, such as the FDA or the EMA, including
2
decisions of such authorities regarding whether and when to approve any drug, device or biological application that may be filed for any product candidates as well as decisions regarding labelling and other matters that could affect the availability or commercial potential of any product candidates, the absence of a guarantee that any product candidates, if approved, will be commercially successful, the future approval and commercial success of therapeutic alternatives, Sanofi’s ability to benefit from external growth opportunities, to complete related transactions and/or obtain regulatory clearances, risks associated with intellectual property and any related pending or future litigation and the ultimate outcome of such litigation, trends in exchange rates and prevailing interest rates, volatile economic and market conditions, cost containment initiatives and subsequent changes thereto, and the impact that global crises may have on us, our customers, suppliers, vendors, and other business partners, and the financial condition of any one of them, as well as on our employees and on the global economy as a whole. While the list of factors presented here is representative, no list should be considered a statement of all potential risks, uncertainties or assumptions that could have a material adverse effect on companies’ consolidated financial condition or results of operations. The foregoing factors should be read in conjunction with the risks and cautionary statements discussed or identified in the public filings with the U.S. Securities and Exchange Commission (the “SEC”) and the Autorité des marchés financiers made by Sanofi, including those listed under “Risk Factors” and “Cautionary Statement Regarding Forward-Looking Statements” in Sanofi’s annual report on Form 20-F for the year ended December 31, 2024 and its other filings with the SEC and the current reports on Form 8-K, quarterly reports on Form 10-Q and annual reports on Form 10-K and other filings with the SEC filed by Blueprint. The forward-looking statements speak only as of the date hereof and, other than as required by applicable law, Sanofi and Blueprint do not undertake any obligation to update or revise any forward-looking information or statements.
3
Exhibit 99.1

Viva Engage Post from Brian Foard to Message to Specialty Care employees
Timing: Monday, June 2, 07:10am CET / 01:10am ET
Important Notices regarding Forward Looking Statements and Additional Information for US Shareholders
Dear colleagues,
Today we signed an agreement to acquire Blueprint Medicines, a global pharmaceutical focused in systemic mastocytosis, a rare immunological disease, and other KIT-driven diseases. Headquartered in Cambridge, MA, Blueprint Medicines has ~650 employees, with the majority sitting in the US. The acquisition includes a rare immunology disease business, Ayvakit (avapritinib) in the US and Ayvakit in Europe, for advanced and indolent mastocytosis, and a promising advanced and early-stage immunology pipeline.
This deal is a strong, strategic fit, for Sanofi and for Blueprint Medicines. We share a deep commitment to the treatment of rare conditions, and immunology and inflammation. AYVAKIT is the first FDA approved therapy for systemic mastocytosis, a rare immunological disease which is characterized by the accumulation and activated aberrant mast cells in bone marrow, skin, the gastrointestinal tract, and other organs.
Like us, Blueprint Medicines believes that strong investment in R&D coupled with patient integration accelerates treatment innovation. They have a broad and compelling pipeline of investigational therapies that align with our own ambitions in Immunology.
A key component of our Take the Lead strategy is making decisions that will deliver long-term, sustainable growth that will benefit all of our stakeholders. It’s through investments like this that we’ll be able to bring more transformational medicines to patients faster.
We expect to close the transaction in Q3 2025. Until then, Blueprint Medicines and Sanofi will continue to operate independently, while integration plans are being developed. We will keep you informed of our progress.
Thank you for all that you do.
With gratitude,
Brian
IMPORTANT: Until the transaction closes, Sanofi employees should not communicate to or with Blueprint employees. All communication will occur through the integration planning team only.
SANOFI 46, avenue de la Grande-Armée, 75017 Paris - Tél.: +33 (0)1.53.77.40.00 - www.sanofi.com
Société anonyme au capital de 2 452 461 656 € - 395 030 844 R.C.S. Paris - Code APE 7010 Z - N°TVA intracommunautaire : FR 88 395 030 844
Exhibit 99.2

Viva Engage Post from Houman Ashrafian to Sanofi R&D Organization
Timing: Monday, June 2, 12:00pm CET / 6:00am ET

Important Notices regarding Forward Looking Statements and Additional Information for US Shareholders
Dear colleagues,
This morning, we announced that we’ve entered into an agreement to acquire Blueprint Medicines, significantly enhancing our position in rare diseases and immunology.
In addition to the acquisition of Ayvakit/Ayvakyt, the only approved disease-modifying medicine for advanced and indolent systemic mastocytosis (ASM & ISM), this strategic acquisition brings an impressive early-stage pipeline that complements our immunology portfolio. The acquisition includes elenestinib, a next-generation medicine for systemic mastocytosis (SM), and BLU-808, an innovative oral wild-type KIT inhibitor with broad potential across immunological diseases. These promising candidates exemplify our commitment to breakthrough science in areas of high unmet need.
The acquisition also includes groundbreaking research in mast cell biology, a critical area of immunology. Mast cells are fundamental players in immune responses, and understanding their role in diseases like SM opens new frontiers for therapeutic innovation. By combining our R&D expertise with Blueprint’s innovative pipeline and scientific capabilities, we’re accelerating our transformation into the world’s leading immunology company.
IMPORTANT: Until the transaction closes, Sanofi employees should not communicate to or with Blueprint employees. All communication will occur through the integration planning team only.
Sincerely yours,
Houman Ashrafian
Head of Research and Development
SANOFI 46, avenue de la Grande-Armée, 75017 Paris - Tél.: +33 (0)1.53.77.40.00 - www.sanofi.com
Société anonyme au capital de 2 452 461 656 € - 395 030 844 R.C.S. Paris - Code APE 7010 Z - N°TVA intracommunautaire : FR 88 395 030 844
Exhibit 99.3
LinkedIn Post:

X Post:


Exhibit 99.4
Acquisition of Blueprint in rare diseases June 2, 2025

Forward-looking statements This communication contains forward-looking statements that are subject to risks, uncertainties and other factors that could cause actual results to differ materially from those implied by the forward-looking statements. Forward-looking statements are statements that are not historical facts and may include projections and estimates and their underlying assumptions, statements regarding plans, objectives, intentions and expectations with respect to future financial results, events, operations, services, product development and potential, and statements regarding future performance. Forward-looking statements are generally identified by the words “expects”, “anticipates”, “believes”, “intends”, “estimates”, “plans”, “will be” and similar expressions. Although Sanofi’s management believes that the expectations reflected in such forward-looking statements are reasonable, investors are cautioned that forward-looking information and statements are subject to various risks and uncertainties, many of which are difficult to predict and generally beyond the control of Sanofi, that could cause actual results and developments to differ materially from those expressed in, or implied or projected by, the forward-looking information and statements. These risks and uncertainties include among other things, unexpected regulatory actions or delays, or government regulation generally, that could affect the availability or commercial potential of the product, or the fact that the product may not be commercially successful, and risks related to Sanofi’s and Blueprint’s ability to complete the acquisition on the proposed terms or on the proposed timeline or at all, including the receipt of required regulatory approvals, the risk that the conditions to the closing of the transaction may not be satisfied, the possibility that competing offers will be made, the risks that the milestones related to the contingent value right will not be achieved, the risk of securityholder litigation relating to the proposed acquisition, including resulting expense or delays, other risks associated with executing business combination transactions, such as the risk that the businesses will not be integrated successfully, that such integration may be more difficult, time-consuming or costly than expected or that the expected benefits of the acquisition will not be realized, risks related to future opportunities and plans for the combined company, including uncertainty of the expected financial performance and results of the combined company following completion of the proposed acquisition, disruption from the proposed acquisition making it more difficult to conduct business as usual or to maintain relationships with customers, employees, manufacturers, suppliers or patient groups, and the possibility that, if the combined company does not achieve the perceived benefits of the proposed acquisition as rapidly or to the extent anticipated by financial analysts or investors, the market price of Sanofi’s shares could decline, as well as other risks related to Sanofi’s and Blueprint’s respective businesses, including the ability to grow sales and revenues from existing products and to develop, commercialize or market new products, competition, including potential generic competition, the uncertainties inherent in research and development, including future clinical data and analysis, regulatory obligations and oversight by regulatory authorities, such as the FDA or the EMA, including decisions of such authorities regarding whether and when to approve any drug, device or biological application that may be filed for any product candidates as well as decisions regarding labelling and other matters that could affect the availability or commercial potential of any product candidates, the absence of a guarantee that any product candidates, if approved, will be commercially successful, the future approval and commercial success of therapeutic alternatives, Sanofi’s ability to benefit from external growth opportunities, to complete related transactions and/or obtain regulatory clearances, risks associated with intellectual property and any related pending or future litigation and the ultimate outcome of such litigation, trends in exchange rates and prevailing interest rates, volatile economic and market conditions, cost containment initiatives and subsequent changes thereto, and the impact that global crises may have on us, our customers, suppliers, vendors, and other business partners, and the financial condition of any one of them, as well as on our employees and on the global economy as a whole. While the list of factors presented here is representative, no list should be considered a statement of all potential risks, uncertainties or assumptions that could have a material adverse effect on companies’ consolidated financial condition or results of operations. The foregoing factors should be read in conjunction with the risks and cautionary statements discussed or identified in the public filings with the U.S. Securities and Exchange Commission (the “SEC”) and the Autorité des marchés financiers made by Sanofi, including those listed under “Risk Factors” and “Cautionary Statement Regarding Forward-Looking Statements” in Sanofi’s annual report on Form 20-F for the year ended December 31, 2024 and its other filings with the SEC and the current reports on Form 8-K, quarterly reports on Form 10-Q and annual reports on Form 10-K and other filings with the SEC filed by Blueprint. The forward-looking statements speak only as of the date hereof and, other than as required by applicable law, Sanofi and Blueprint do not undertake any obligation to update or revise any forward-looking information or statements. Blueprint Medicines, Ayvakit, Ayvakyt, and associated logos are trademarks of Blueprint Medicines Corporation.

Additional information for US shareholders of Blueprint The tender offer for the outstanding shares of Blueprint Medicines Corporation (“Blueprint”) common stock referenced in this communication has not yet commenced. This communication is for informational purposes only and is neither an offer to purchase nor a solicitation of an offer to sell shares of Blueprint, nor is it a substitute for the tender offer materials that Sanofi and its acquisition subsidiary will file with the U.S. Securities and Exchange Commission (the “SEC”) upon commencement of the tender offer. At the time the tender offer is commenced, Sanofi and its acquisition subsidiary will file tender offer materials on Schedule TO, and Blueprint will file a Solicitation/Recommendation Statement on Schedule 14D-9 with the SEC with respect to the tender offer. The tender offer materials (including an Offer to Purchase, a related Letter of Transmittal and certain other tender offer documents) and the Solicitation/Recommendation Statement will contain important information. HOLDERS OF SHARES OF BLUEPRINT ARE URGED TO READ THESE DOCUMENTS WHEN THEY BECOME AVAILABLE BECAUSE THEY WILL CONTAIN IMPORTANT INFORMATION THAT BLUEPRINT STOCKHOLDERS SHOULD CONSIDER BEFORE MAKING ANY DECISION REGARDING TENDERING THEIR SHARES. The Offer to Purchase, the related Letter of Transmittal and certain other tender offer documents, as well as the Solicitation/Recommendation Statement, will be made available to all holders of shares of Blueprint at no expense to them. The tender offer materials and the Solicitation/Recommendation Statement will be made available for free at the SEC’s web site at www.sec.gov. Additional copies may be obtained for free by contacting Sanofi’s Investor Relations Team at investor.relations@sanofi.com or on Sanofi’s website at https://www.sanofi.com/en/investors. In addition to the Offer to Purchase, the related Letter of Transmittal and certain other tender offer documents, as well as the Solicitation/Recommendation Statement, Sanofi files annual and special reports and other information with the SEC and Blueprint files annual, quarterly and special reports and other information with the SEC. You may read and copy any reports or other information filed by Sanofi and Blueprint at the SEC public reference room at 100 F. Street, N.E., Washington D.C. 20549. Please call the Commission at 1-800-SEC-0330 for further information on the public reference room. Sanofi’s and Blueprint's filings with the SEC are also available to the public from commercial document-retrieval services and at the website maintained by the SEC at www.sec.gov.
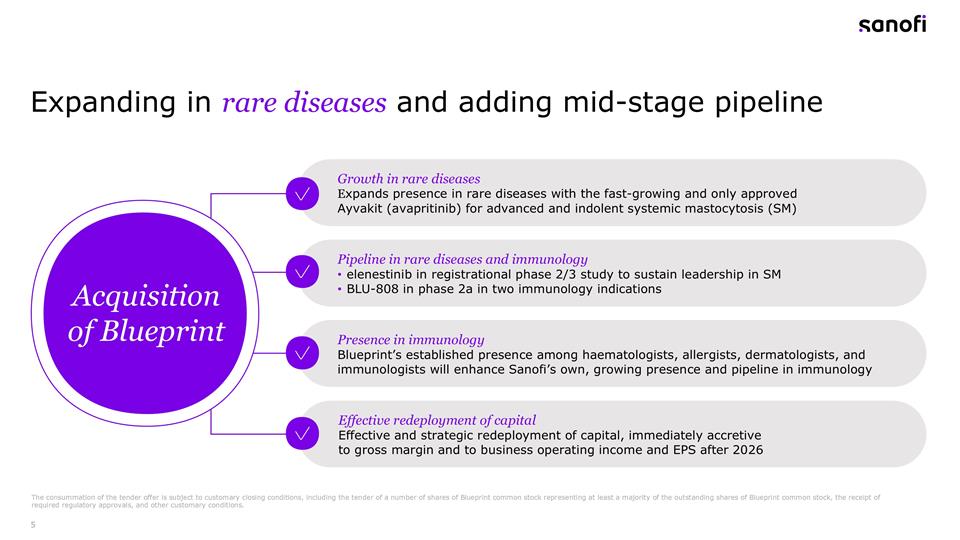
Expanding in rare diseases and adding mid-stage pipeline Growth in rare diseases Expands presence in rare diseases with the fast-growing and only approved Ayvakit (avapritinib) for advanced and indolent systemic mastocytosis (SM) Pipeline in rare diseases and immunology elenestinib in registrational phase 2/3 study to sustain leadership in SM BLU-808 in phase 2a in two immunology indications Presence in immunology Blueprint’s established presence among haematologists, allergists, dermatologists, and immunologists will enhance Sanofi’s own, growing presence and pipeline in immunology Effective redeployment of capital Effective and strategic redeployment of capital, immediately accretive to gross margin and to business operating income and EPS after 2026 Acquisition of Blueprint The consummation of the tender offer is subject to customary closing conditions, including the tender of a number of shares of Blueprint common stock representing at least a majority of the outstanding shares of Blueprint common stock, the receipt of required regulatory approvals, and other customary conditions.
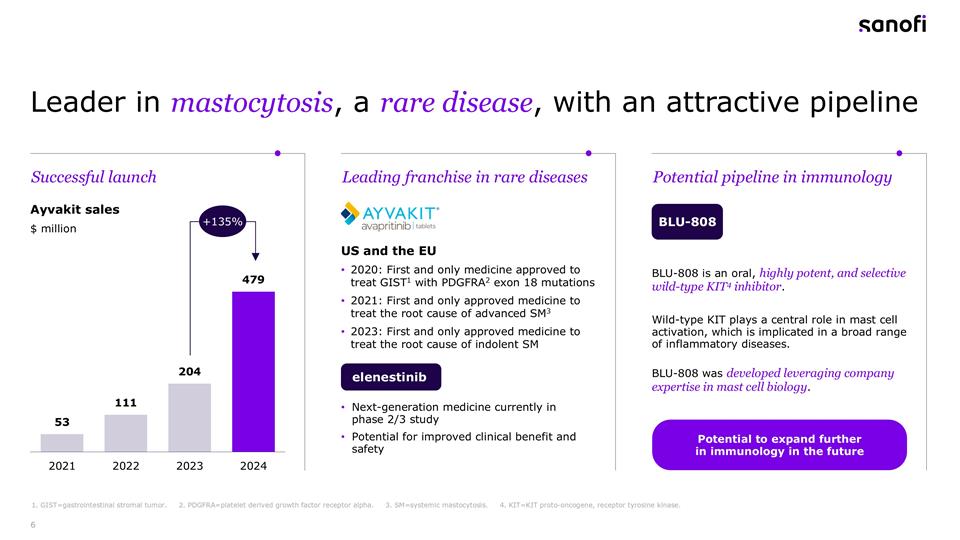
Leader in mastocytosis, a rare disease, with an attractive pipeline 1. GIST=gastrointestinal stromal tumor. 2. PDGFRA=platelet derived growth factor receptor alpha. 3. SM=systemic mastocytosis. 4. KIT=KIT proto-oncogene, receptor tyrosine kinase. Ayvakit sales BLU-808 $ million US and the EU 2020: First and only medicine approved to treat GIST1 with PDGFRA2 exon 18 mutations 2021: First and only approved medicine to treat the root cause of advanced SM3 2023: First and only approved medicine to treat the root cause of indolent SM Next-generation medicine currently in phase 2/3 study Potential for improved clinical benefit and safety Successful launch Potential pipeline in immunology Leading franchise in rare diseases +135% elenestinib BLU-808 is an oral, highly potent, and selective wild-type KIT4 inhibitor. Wild-type KIT plays a central role in mast cell activation, which is implicated in a broad range of inflammatory diseases. BLU-808 was developed leveraging company expertise in mast cell biology. Potential to expand further in immunology in the future

Mastocytosis: a rare and debilitating disease with Ayvakit as the first and only approved medicine for indolent forms Systemic mastocytosis patients include two key sub-sets Indolent systemic mastocytosis Patients have normal life expectancy but debilitating allergic symptoms, including: Healthy mast cells play a key role in immune-inflammatory response Mutation in KIT D816V aberrantly activates mast cells resulting in systemic mastocytosis Over-activation of mast cells results in range of “allergic” symptoms Ayvakit treats systemic mastocytosis through inhibition of KIT D816V KIT mutation drives mast cell over-activity Mast cell over-activity drives ‘allergic’ symptoms 1 2 Biology of systemic mastocytosis and Ayvakit1 Patient experience Advanced systemic mastocytosis Leads to loss of organ function and reduced life expectancy diagnosed SM patients in US claims data in 20242. Only a fraction being treated with Ayvakit today 25k 1. Avapritinib versus placebo in indolent systemic mastocytosis, NEJM Evidence. 2. Internal Blueprint analyses of US claims data at the end of 2024. Headache Gastrointestinal Itching Difficulty breathing

Financial overview and timelines Purchase price Financial considerations Timing and integration Offer price at $129.00 per share in cash at closing, equating to $9.1bn One non-tradeable contingent-value right per share with two potential milestone payments as follows: $2 per share contingent on a clinical development milestone for BLU-808 $4 per share contingent on a regulatory milestone for BLU-808 Immediately accretive to gross margin Accretive to business operating income and EPS after 2026 Established presence among haematologists, allergists, dermatologists, and immunologists will enhance Sanofi’s own, growing presence and pipeline in immunology Financing with available cash resources and proceeds from new debt Expected to close in Q3 2025 Work closely on combination model to maintain momentum
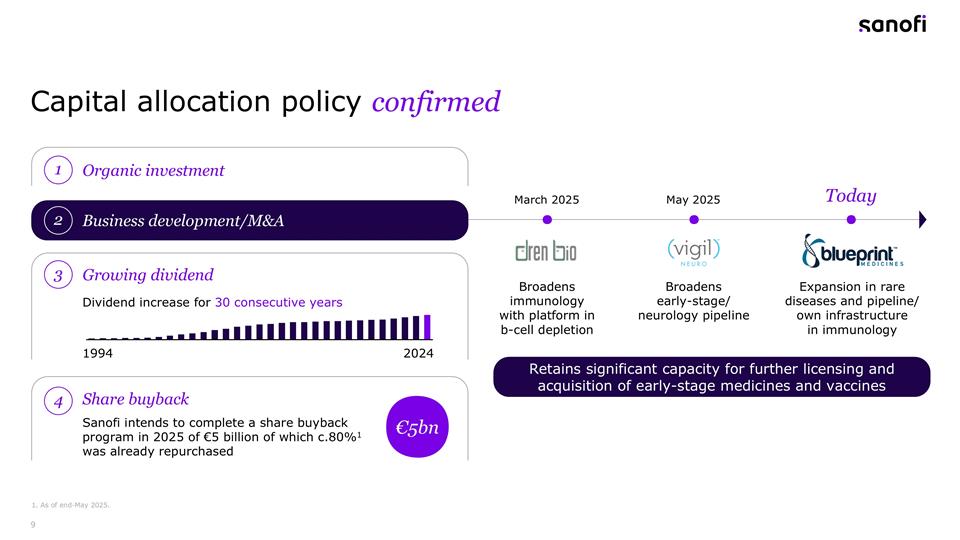
Capital allocation policy confirmed 1. As of end-May 2025. 3 Growing dividend Dividend increase for 30 consecutive years 1994 2024 Expansion in rare diseases and pipeline/ own infrastructure in immunology Broadens early-stage/ neurology pipeline Broadens immunology with platform in b-cell depletion May 2025 March 2025 Today Organic investment 1 Business development/M&A 2 Share buyback 4 €5bn Sanofi intends to complete a share buyback program in 2025 of €5 billion of which c.80%1 was already repurchased Retains significant capacity for further licensing and acquisition of early-stage medicines and vaccines
Expanding in rare diseases and adding mid-stage pipeline Growth in rare diseases Expands presence in rare diseases with the fast-growing and only approved Ayvakit (avapritinib) for advanced and indolent systemic mastocytosis (SM) Pipeline in rare diseases and immunology elenestinib in registrational phase 2/3 study to sustain leadership in SM BLU-808 in phase 2a in two immunology indications Presence in immunology Blueprint’s established presence among haematologists, allergists, dermatologists, and immunologists will enhance Sanofi’s own, growing presence and pipeline in immunology Effective redeployment of capital Effective and strategic redeployment of capital, immediately accretive to gross margin and to business operating income and EPS after 2026 Acquisition of Blueprint The consummation of the tender offer is subject to customary closing conditions, including the tender of a number of shares of Blueprint common stock representing at least a majority of the outstanding shares of Blueprint common stock, the receipt of required regulatory approvals, and other customary conditions.
Q&A session To ask a question Click on the Raise hand icon Check your audio device is well connected Raise and lower your hand: dial *9 Unmute and mute your microphone: dial *6 By phone Email us: investor.relations@sanofi.com Any problems? By zoom

Exhibit 99.5

Exhibit 99.6
02-Jun-2025
Sanofi (SNY)
Investor Relations Call: Acquisition of Blueprint Medicines by Sanofi
Thomas: Good morning, everyone. This is Thomas Kudsk Larsen from the Sanofi IR team. Thank you for joining us today to discuss Sanofi’s proposed acquisition of Blueprint Medicines that significantly strengthens our position in rare diseases and which represents a major step forward in our commitment to patients with rare and often debilitating diseases like mastocytosis. We plan to spend between 30 and 45 minutes today, depending on the number of questions. As usual, you can find the slides on sanofi.com. Please turn to slide 3 and 4. Here we have the usual forward looking statements and a new slide specifically for today. We would like to remind you that information presented in this call contains forward looking statements which are subject to substantial risks and uncertainties that may cause actual results to differ materially. We encourage you to read both disclaimers in our presentation. In addition, we refer you to our 2024 Form 20-F on file with the US SEC and our French registration document for description of these risk factors. With this, I’ll hand you over to our CEO, Paul.
Paul: Well, thanks, Thomas and let me also welcome everyone. On our side, we have Brian, François and Houman to present and help with the Q&A. On slide 5, let me outline the four key pillars of this acquisition. First, we’re significantly expanding our rare disease portfolio through Blueprint’s Ayvakit, the only approved therapy for both advanced and indolent systemic mastocytosis. Second, we’re gaining mid-stage pipeline, including elenestinib in registrational lifecycle management development in mastocytosis and BLU-808 in immunology. Third, this acquisition brings us an established presence among key specialist physicians complementing our own growing presence and pipeline in immunology. And fourth, this acquisition will be immediately accretive to our gross margin with positive impacts on business, operating income and EPS expected after 2026. Before I hand over to Brian, I would also like to welcome the Blueprint colleagues to Sanofi as we look forward to working with you on advancing your unique knowledge of rare diseases, specifically mastocytosis and immunology. Let me hand over to Brian to cover more details.
Brian: Thanks, Paul. The growth trajectory of Ayvakit has been impressive, with sales reaching nearly $479 million in 2024, representing substantial growth over 2023. Q1 2025 was $149 million. A great base for continued fast growth in 2025, supporting the indications Blueprint already has in the marketplace. This growth is driven by more patients being treated and is a strong foundation for the future. This success demonstrates both the significant unmet need in mastocytosis and Blueprint’s excellent commercial execution. Looking forward, elenestinib represents a potential next generation treatment, while BLU-808 offers exciting potential in immunology indications. This portfolio gives us a clear leadership position in mastocytosis treatment and optionality in immunology. As we move to the next slide, slide 7 let’s take a moment to better understand mastocytosis a rare and debilitating disease that is highly symptomatic. It’s caused by a mutation that leads to overactivation of mast cells resulting in a range of allergic symptoms. It is very well-defined biology and amenable to a companion diagnostic. Our focus at Sanofi already is driven through our rare diseases and in immunology. Ayvakit is the first and only approved medicine that treats the root cause of both advanced and indolent forms of systemic mastocytosis. Data suggest that there are approximately 25,000 diagnosed patients with systemic mastocytosis in the US alone, representing a significant opportunity for growth with increased disease awareness and diagnosis both in the US and as Ayvakit is launched outside of the US for ISM. Now let me hand over to François for the financials.
François: Thanks, Brian. For your review turning to the transaction details, we are offering $129 per share in cash, valuing Blueprint at $9.1 billion. Additionally, shareholders would receive contingent value rights worth up to $6 per share tied to specific clinical trials and regulatory milestones for BLU-808. The transaction will be financed through a combination of available cash and new debt. The proposed transaction, sorry, the proposed transaction is subject to customary closing conditions and is expected to close in the third quarter of 2025. Moving to the next slide. This acquisition aligns perfectly with our capital allocation strategy. We remain committed to our dividend policy having increased our dividend for 30 consecutive years. The Blueprint acquisition, along with our recent acquisitions in immunology and neurology, demonstrate the focus on strategic M&A to complement our organic investments. Importantly, we retain significant capacity for further business development, particularly in early stage medicines and vaccines across our core disease areas. To summarize this acquisition of Blueprint Medicines significantly strengthen our position in rare diseases, particularly in systemic mastocytosis. We are gaining an attractive pipeline, including elenestinib and BLU-808 on a fast growing approved medicine in Ayvakit. As well as an established presence among key specialist physicians as a great platform for Sanofi’s own and growing pipeline in immunology. This transaction represents an effective redeployment of capital with immediate accretion to our gross margin and expected positive impacts on business, operating income and EPS after 2026. Let me now hand back to Paul for Q&A.
Paul: Well, thank you, François. We’ll now open the floor for questions. I will not ask to limit to one as that strategy has never worked for me. If you’re joining us via Zoom, please use the “raise hand” feature to indicate you’d like to ask a question. For those on the phone, you can raise your hand by dialing star nine and unmute yourself by dialing star six when called upon. Marie will announce the next person in line.
Operator: Yes, the next question and first question is from Luisa Hector from Berenberg. Luisa.
Louisa: Good morning. Thank you very much for the call and a nice deal. I’ll try and keep it to one question. Can you say a little bit more about what you have assumed in your valuation here? You have the lead product on the market. Is that really about the indications that are on label now? Have you assumed further indications and any pipeline? Thank you.
Paul: Okay. François, maybe to kick off with the valuation of the deal with some headlines and Brian, you will talk about the indication.
François: Yes, the valuation Luisa thank you for the question. So, you have seen the company was an independent company and they have met their own guidance as well. So we feel totally comfortable with what they have done. Obviously, we have run our own model and assumed a certain number of synergies as well. But globally, we are comfortable with whatever Blueprint has published in the past.
2
Paul: Okay. So, Brian, would you like to answer now?
Brian: Yeah, I think, Luisa a great question. And I think as you look at this company, it’s quite impressive what they’ve done already. But as we have looked at them, we saw both in the lead asset that’s on the marketplace. Of course, this is just getting started. It’s very early into the launch phase. So we’ve seen from our valuation, we look at the potential of that asset over the long period of time. And if that first has to start with, it’s a really extremely low advance therapy penetration into the marketplace thus far. So in this deal, we looked at the growth potential for that asset. First and foremost. Number one, we look at the follow-on asset in elenestinib with the potential for differentiation there, but sustainability, long-term leadership sustainability in ISM of course. And then of course, we looked at their BLU-808 as well. We looked at that asset in immunology indication. So those were the by and large how we thought about this deal longer-term.
Paul: Okay. Thank you. Thank you, Luisa, Brian. Next question, please.
Operator: Yes, next question is from Matthew Weston from UBS. Matthew.
Matthew: Thank you. I guess trying to put the transaction in numbers to understand the valuation that you’re comfortable paying. Brian just flagged the whole rationale being the sustainable benefit of the franchise in mastocytosis. So can you give us the peak sales potential that you see for that sustainable mastocytosis franchise going forward ? And then also on BLU-808 and we understand whether you see potential in a larger breadth of indications than Blueprint was currently prosecuting or whether or not you see it limited to the two indications that they currently have in it?
Paul: So thank you, Matthew. It’s a bit early to get into sharing peak year sales projections with us, of course, announcing the signing. And François mentioned that Blueprint themselves have published some projections. You know, we work many different scenarios. You’ll appreciate we are very competent in rare diseases, perhaps one of the absolute best. We’re ambitious for that. It’s too early for us to share the numbers. In terms of the longevity of the franchise, there is an expectation, modest, that we can move from Ayvakit to elenestinib and that’s a very reasonable assumption. As for BLU-808, it’s early. I mean, it’s one of those things that scientifically could be very compelling. But, you know, I don’t need to remind anybody, particularly our own people in-house, that, you know, there are many things that can happen between a medicine and getting to market or not. But you’d appreciate that we like all of the assets, but some have a different level of risk.
Operator: Next question is from Richard Vosser from JPMorgan. Richard.
Richard: Thanks for taking my question. You mentioned synergies. How should we think about the level of synergies relative to Blueprint’s OpEx? Could we anticipate most of their SG&A being saved? Some thoughts there? And just on the duration of AYVAKIT, it looks like the patents go to 2034, but there’s a crystalline patents 2040. So should we be thinking about 2040? And I’m assuming that with orphan drug, there’s no direct negotiation issues. But if you could just confirm that, that would be useful. Thanks very much.
3
Sanofi: Well, first and foremost, Richard, thank you so much for your question. And I’ll start with the second one that we won’t really comment right now at this particular point on IP. But I will go back to I want to go back to your first part of your question was, as we said, I think if you look at the success of Blueprint, this is why we looked at these folks and why at the end of the day, we’re actually doing this deal is. If you think about where they are today, their call points uniquely fit us perfectly. So think about our pipeline that is being built right now with amlitelimab, lunsekimig, TL1A. The call points for those assets are going to be dermatologist, allergist, GIs which is exactly where Blueprint is today. So it is going to be a natural build around these individuals. So right now we’re not thinking about the synergies of this. Right now we’re thinking about the opportunity for growth beyond what Blueprint has already done. And as we grow our pipeline, which is going to be extremely synergistic to the call points that they are already in place with.
Paul: Yeah. And maybe I add actually that they’ve done an incredible job. I think we bring a global reach in rare that is pretty much unparalleled. I think we’re looking forward to learning from them in some areas and we’re also looking forward to adding to it. So, you know, of course, with way too early, we’re get into the planning jointly with them and we want to learn from them to do the very best job. I think you all appreciate that in rare and specialty in particular rare, we’re not talking about large organizations. We’re talking about meaningful roles, avoiding duplication and making sure that people want to be part of the project. And I think we’re excited about that. We’ll sit down with them, we’ll work through that. They have a really impressive team, so we’ll get to know them better and decide from that.
Operator: The next question is from James Quigley from Goldman Sachs. James.
James: Hello. Can you hear me okay? Apologies about that. Just one quick question from my side in terms of the CVR. if could give us a little bit more details on what clinical milestones are that have to be hit to hit both the two-dollar and four-dollar CVR. And second of all, just a quick one on timing as well. I think the impression I got previously was that maybe you’re more focused on earlier stage deals as opposed to commercial stage deals. Obviously, relative you wouldn’t be looking at deals in terms of itepekimab have been a timing there. But give us an overview of what this now means for your M&A strategy and where the focus would be from here as well. Thank you.
Sanofi: Okay. I think I got most of that. Just I think it’s actually a good moment to just be very clear with everybody, those that are close to how these things work know that they’re a long time in the making. And so this has been going on long before the itepekimab update of last week. The first thing. Secondly, I don’t think we’re commenting on the CVR milestones and where they are, but I think both sides are comfortable that we did something that was appropriate for value creation on our side and respect to the legacy of what they’ve built from their side. We’re comfortable with that. Was one of the parts of question. Oh, yeah. And on the deals, you know, we sound a bit like a broken record sometimes then we would just I think the things that we’ve said, you know, a large amount of times is €2 billion to €5 billion remain opportunistic over the last year or so because we wanted to be clear with people. We said we’ll keep it single digit if it’s something that’s a little bit more exciting for us but is within our TAs. We said we wouldn’t rush
4
to go and spend high single digit away from where we needed to be. So we tried to stay very disciplined on that. The question on the early stage preclinical, you’ve seen some little bits and pieces that we’ve done recently. You’ll see more up ahead because we have great belief in our mid late stage pipeline. Let’s be candid, we had a few bumps, but we are still see very clear path forward. The things we announced earlier this year. But I still think we are a little bit underweight in first-in-class, best-in-class, preclinical Phase 1. You’ll still see us doing that. So I think you should that nothing really changed for us. I think that’s consistent with what we said and how we feel, which is the best way to think.
François: And maybe to complement, François speaking, really to complement what Paul said as well. As you know, we had the very low level of debt we were just a few weeks ago. We were even with a net cash positive situation, even after this transaction and the two transaction that we announced earlier this year, we will still be left with a net debt to EBITDA ratio, post Blueprint acquisition, of around one in terms of net debt to EBITDA. That gives us flexibility even to go further. Obviously, given that we can go significantly further, even meeting our objective of retaining or double everything. And we will remain disciplined anyway. The point is not to just go and make acquisition for the sake of making acquisition, but it’s about making sure that we find relevant acquisition from a strategic point of view, from a scientific point of view, having differentiating assets from the financial point of view with the proper retail for all shareholders.
Paul: Yeah. François well said. Actually the rate limiting step given you know, I look back over the last five years, Principia was perhaps one of our biggest acquisitions. It’s a while ago now the rate limiting step for us has always been the right science with the right fit and the right growth potential and opportunity for us. We’ve been very disciplined on that. In fact, François conducted a review not long after he joined the company to go back and look at how we’ve allocated capital and did we make sense with what we did? Now that we can look back with hindsight, and of course he wasn’t involved in any of these things and gave us a thumbs up that we’re good custodians of how we deploy and we feel exactly the same going into this.
François: Just to give more color, to be more specific, if you look at what Sanofi has done over the last 10 years. Sanofi has invested €40 billion, 4-0, €40 billion in M&A on BD, out of which we have lost, sorry for that. Only if I can say only 10%, €4 billion, which is not a lot essentially, by the way, and most entirely in oncology. Hence, I mean, the decisions that have been made recently regarding oncology and the remaining €36 billion have created a lot of value. And Paul was mentioning Principia to public information. We bought it to €3 billion. If you look at the value of this acquisition, it was probably three to four times by now, and probably even more in the future. So I think that Sanofi has proven to be very disciplined in terms of M&A and BD strategy, providing good returns to its shareholder.
Operator: Okay. Next question. Emmanuel Papadakis from Deutsche Bank. Emmanuel.
Emmanuel: Merci and bonjour. Thank you for taking the questions. So perhaps a couple, if I could, it sounds like BLU-808 was a reasonably significant component of the value proposition. Could you just talk a little bit about which indications, if any, you’re most interested? Clearly, some of those do, to an extent, overlap with your existing assets or pipeline portfolio. And then just a quick follow-up on that comment on oncology—Blueprint does obviously have a significant early stage oncology effort. Could you just talk about you haven’t mentioned that or could you just talk about the extent to which, if any, that played a role in your interest in the acquisition and what it signals about your future intent in returning to that space? Thank you.
5
Sanofi: Emmanuel good morning and thank you for the question. It’s a bit early to comment in any detail about BLU-808 suffice to say that over the last 10 years there’s been a recrudescence of interest in mast cell biology, both in wild-type form and in the mutated Da816v variant. And we will be looking broadly, as Brian has commented, in multiple organ areas as we move forward. That matches what’s now known about mast cells. And to your oncology comment, it’s really important to say that we’re excited about the Blueprint team joining us and working together, as Paul said, learning from them and sharing insights around the biology not only of the mast cells but oncology. Remember, we have a creditable early oncology program and our internal team are looking forward to working in multiple areas with Blueprint, including the greatest. Thanks for the question.
Paul: Yeah. And you did also mention about where we are pushing the value and you know, the the key driver who is the lead asset approved asset that’s growing significantly. And kudos again to Blueprint team. Elenestinib and running opportunity to grow through the 30s is a big deal, as you’d appreciate. And we’re not going to talk about the relative weightings of where we deployed the capital in the deal. But, you know, BLU-808 is early . It could be exciting. But of course, like any asset at that stage, there’s risk. So you should imagine proportionately less value in that.
Operator: Next question is from Peter Verdult from BNP. Peter.
Peter: Yeah. Good morning, Peter Verdult, BNP. Three quick questions. Firstly, Houman or Paul, in terms of Trump administration proposals to price control rare disease assets that have more than one indication, how concerning is that to you as it relates to the whole rare disease portfolio? Two, for François you talked about flexibility and comfort being one times post the deal. Could I push you in terms of where would you be uncomfortable in terms of leverage at two times three times just some sort of idea of firepower and capacity? And then lastly, Paul, just speaking to investors, you know, there’s a lot of focus on being able to navigate the dupixent LOE. I think people are very comfortable with tolebrutinib, €2 billion to €3 billion achieved, obviously goes to the sidelines. So just, you know, there’s a perception of rising binary risk around the year-end readout for amlitelimab. So just would like your latest thoughts and confidence that that profile deal seeking for amlitelimab will be reached. Thank you.
Brian: Yeah, I think the first question, Peter, as you mentioned, is in reference to the current administration of reference around price. You know, look, we work with all administrations around the world. We’ve been doing this for a very, very long time. We’ll see how this particular situation plays out. But we feel like we’re in one of the best positions, I think, as it relates to our peers, as it relates to price around the world, specifically, as it relates to rare diseases. Again, this is an area where we have deep subject matter expertise. But again, administration by administration, you know, brings on new sets of challenges and we’ll work through them day by day. But we feel like we’re in a very good position as it relates to price.
Operator: Thank you. François. Any comment from you?
6
François: Yeah. On the rating as an indication because it’s not up to us to decide. It’s up to the rating agencies, but to retain our AA rating, we cannot really exceed the two as a net debt to EBITDA ratio. I just want to make sure that this is not a guidance, but it is an indication.
Paul: Okay. Thank you. And to your last point Peter it’s quite interesting really for us because I said we’ve had a bit of a bumpy year in terms of some of the readouts. And what I mean by that is, you know, we looked at the amlitelimab data in asthma. We know we have a medicine we know will go forward, but we didn’t meet the primary endpoint. It didn’t change for us our expectations, frankly. Same with the old TNF. You know, we never intended to develop the asset to win in psoriasis. It was all about going there to get a safety readout first before we went on to RA. And that’s a bit more interesting, and that’s later this year, but still on track, by the way. But itepekmab, frankly, It’s a bit of a confounder only because we’ve been in this situation before and many of us have in the industry. And so we’ll spend a bit more time going much deeper into what we think is the driver in [indiscernible], too. We have some early thoughts. We’ll do that with our partners, Regeneron. And we’ll see if we have a path forward or whether we need to do a little bit more work. It is too early to call it and it’s too early I think, to use your words, to put it on the sidelines. The last thing would be the amli readout. I have to say, irrespective of the other things which are all fabulous, you know, amlitelimab in AD you know, is the thing that is, you know, perhaps one of the biggest value drivers this year in terms of readouts. No surprise. So we look forward to that expectantly and we’ll see where we get to. So I recognize that the year has been, as I said, bumpy, but actually the underlying sentiment, internally at least, is on track to do what we need to get done. And we look forward to the amli readout later this year.
Operator: And we don’t have any raised hand. Does anyone has any question? We have one from Jaime Escribano. Jaime, if you want to ask your question.
Jaime: Hello. Hi. Can you hear me?
Operator: Yes.
Jaime: Yes. Okay. Thank you. Yeah. My question was to clarify the patent cliff for the main product of Blueprint. So is it 2040, or is it more 2034? Because the product was launched in 2021. So just to make sure what’s the visibility on earnings will have. And the second question would be we read Blueprint pointing to $2 billion peak sales. The question would be, what would be the minimum that you would like to see on this product in order to consider this to be a good deal? Of course, then you have the other two compounds in the pipeline. But just to have a sense on your thresholds? Thank you very much.
Paul: Okay. So, Brian, maybe some first thoughts from you.
Brian: Yeah, I’ll say again, as we said before, we won’t comment fully on IP other than to say that we’re extremely comfortable with the IP based upon what we’ve seen so far, but we’ll comment on it in more detail as it close. As it relates to the IP of the asset of the lead asset with Ayvakit. As it relates to peak sales, again, we mentioned it, Blueprint put out some peak to peak sales guidance, if you will. That’s all public. We are not validating those peak sales here on this
7
particular call. We’ll give a bit more details, I think it close as well as it relates to peak sales. But again, as I said it first and foremost, it all goes back to the potential we see here is the significant growth potential that we see as it relates to significant amount of patients undiagnosed and incredible therapy that’s already on the marketplace, follow-on asset that actually could extend the our leadership in that particular space over a long period of time, and again potential follow-on assets in BLU-808.
Paul: I mean I covered it today. I’m not sure there’s much more to add. Operator, any more questions?
Operator: Any other question?
Paul: Well, thank you all for your attention. We’re excited about the future of Sanofi in rare diseases with the addition of Blueprint Medicines and look forward to updating you on our progress over the coming months. With that, we’ll end the call. Thank you.
8
